Press release
Butamirate Impurity Standards: A Commitment to Purity and Safety in Pharmaceutical Excellence
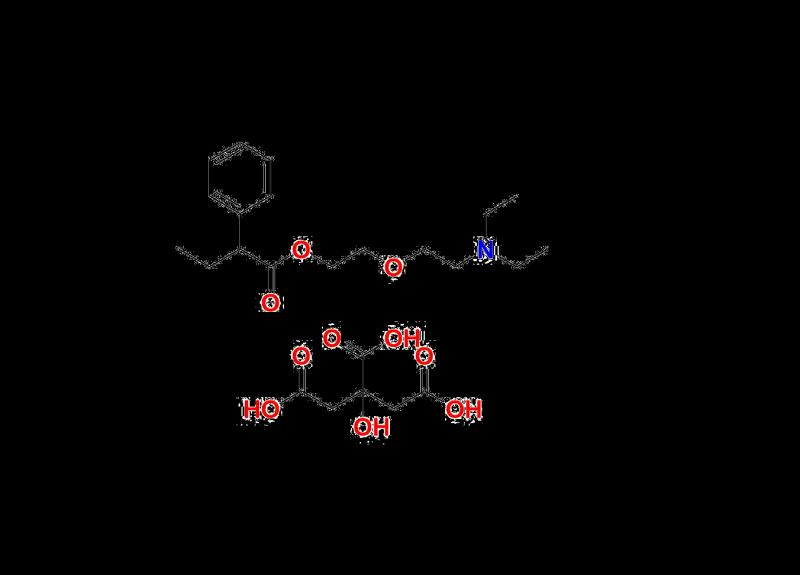
March 19, 2025 - Pune, India | Butamirate, a widely recognized active pharmaceutical ingredient (API), plays a pivotal role in formulating effective cough suppressants, alleviating respiratory distress and promoting patient well-being. In the production of Butamirate, the presence of impurities poses a critical concern, as they may affect the efficacy, safety, and stability of pharmaceutical products. Impurities can arise during synthesis, storage, or processes involving raw materials and intermediates. Therefore, the development and use of robust impurity standards are essential. These standards assist manufacturers in identifying and quantifying impurities and ensure compliance with global regulatory guidelines issued by authorities such as the US FDA and EMA.to know more about category : https://aquigenbio.com/products/impurity-standards/butamirate/
The Crucial Role of Butamirate Impurity Standards in Pharma
The pharmaceutical industry is driven by its commitment to upholding rigorous quality standards, and Butamirate is no exception. Impurity profiling is an integral part of the development and validation processes that ensures high standards of purity, thereby guaranteeing therapeutic safety. To this end, impurity reference standards for Butamirate play a key role by enabling manufacturers and researchers to accurately detect and limit impurities in formulations.
For example, the identification of Butamirate Impurity 1 requires precise analytical measurement tools and standardized protocols to prevent its accumulation in final pharmaceutical products. Similarly, addressing Butamirate Impurity 2, another potential contaminant, demands equal precision to ensure that products meet acceptable limits established by pharmacopeias and global standards. These efforts are not merely technical processes but safeguards for patient health, reinforcing the overarching objectives of the pharmaceutical industry.
Butamirate Citrate: Ensuring Efficacy and Compliance
Butamirate citrate, a commonly used salt form of the API, is a prime choice in the preparation of antitussive (cough suppressant) medications. This derivative is valued for its superior solubility and therapeutic effectiveness, making it a preferred inclusion in global cough formulations. As with the core API, ensuring the purity of Butamirate citrate involves rigorous impurity analysis. By leveraging scientifically sound methods and validated reference standards, pharmaceutical manufacturers can consistently produce high-quality Butamirate citrate formulations that comply with internationally accepted safety benchmarks.
To know more about products:-
https://aquigenbio.com/product/butamirate-citrate/
https://aquigenbio.com/product/butamirate-impurity-1/
https://aquigenbio.com/product/butamirate-impurity-2/
Comprehensive Research and Analytical Excellence
Advancements in analytical techniques have significantly enhanced the pharmaceutical industry's ability to detect and manage even trace levels of impurities in Butamirate formulations. Cutting-edge technologies, such as high-performance liquid chromatography (HPLC), gas chromatography (GC), mass spectrometry (MS), and nuclear magnetic resonance (NMR), enable precise detection of impurities and their quantification at exceptionally low levels. These tools are indispensable in meeting regulatory expectations for quality control while continuously striving to mitigate risks associated with impurity-related toxicity.
By embracing these advanced analytical techniques, drug manufacturers can ensure that their Butamirate-based products maintain high levels of purity, stability, and therapeutic efficacy. Additionally, consistent adherence to impurity standards facilitates uninterrupted market access, increases consumer trust, and prevents costly recalls due to non-compliance.
Impurity Standards: The Backbone of Regulatory Compliance
The pharmaceutical industry today operates within a dynamic regulatory landscape where impurity management forms the backbone of compliance strategies. Regulators worldwide mandate that impurity levels in drugs be scientifically justified and fall below predetermined thresholds. These mandates apply specifically to APIs like Butamirate, where the presence of impurities can have serious implications for product safety and efficacy.
Pharmaceutical manufacturers are thus expected to establish validated impurity standards as part of their drug development programs. Systematic impurity profiling of Butamirate Impurity 1, Butamirate Impurity 2, and other potential contaminants serves as the cornerstone of such compliance efforts. This rigorous focus on quality and patient safety reinforces the importance of adopting high standards for active ingredient production and the critical role of impurity testing.
Conclusion
In an era where patient safety and regulatory standards are of paramount importance, the pharmaceutical industry's focus on impurity profiling underscores its unwavering commitment to quality and compliance. By leveraging advanced analytical approaches, validated reference standards, and adhering to stringent guidelines, manufacturers ensure the purity of Butamirate-based formulations, safeguarding patient health and enabling therapeutic efficacy.
A clear and comprehensive understanding of Butamirate impurities set the foundation for producing safe and reliable cough suppressant medications. Pharmaceutical researchers and manufacturers alike continue to push boundaries to ensure that stringent benchmarks for quality and safety are consistently met, marking an important step forward in global healthcare.
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
Aquigen Bio Sciences is India's leading provider of high-quality impurity standards, specializing in solutions for Butamirate and its derivatives. With a proven track record in supplying validated references for pharmaceutical impurity profiling, Aquigen Bio Sciences supports researchers and manufacturers in achieving unmatched precision and accuracy. Leveraging state-of-the-art technologies and deeply rooted expertise, the company has become a trusted partner in maintaining pharmaceutical quality, compliance, and innovation across the industry. Aquigen Bio Sciences is the definitive resource for manufacturers seeking reliable impurity standards, delivering impurity standards that reinforce patient safety.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Butamirate Impurity Standards: A Commitment to Purity and Safety in Pharmaceutical Excellence here
News-ID: 3923715 • Views: …
More Releases from Aquigen Bio Sciences

Elevate Pharmaceutical R&D with Aquigen BioSciences' Precision‐Grade Flibanser …
Flibanserin Impurity B is a reference standard used in pharmaceutical research and development. It is primarily applied during the analysis and validation of drug substances to identify, quantify, and control impurities that may be present in the final product. This impurity is associated with the parent compound, Flibanserin, a medication approved for the treatment of hypoactive sexual desire disorder (HSDD) in premenopausal women.
Aquigen BioSciences offers Flibanserin Impurity B as a…

Estradiol Valerate EP Impurity A - Premium Reference Standard for Analytical Dev …
Estradiol Valerate EP Impurity A is a high-quality reference standard designed to meet the stringent requirements of pharmaceutical research, method validation, and quality control processes.
Explore Estradiol Valerate EP Impurity A :
https://aquigenbio.com/product/estradiol-valerate-ep-impurity-a/
Manufactured and characterized with precision, this impurity standard supports laboratories and manufacturers in achieving consistent, reliable, and reproducible results in critical analytical workflows.
With its exceptional purity and accurate characterization, Estradiol Valerate EP Impurity A plays a vital role…

High-Purity N-Nitroso Betahistine D3 for Precise Pharmaceutical Analysis | Deute …
Product Overview
N-Nitroso Betahistine D3 is a premium deuterated nitrosamine impurity standard, specifically developed for precise analytical testing in pharmaceutical laboratories. This reference standard is widely used for analytical method development, validation, and quality control processes to meet stringent regulatory guidelines. With exceptional purity, complete documentation, and reliable traceability, it is ideal for research, development, and compliance applications.
https://aquigenbio.com/product/n-nitroso-betahistine-d3/
Key Features and Benefits
Deuterated Design for Precision: The incorporation of deuterium improves mass spectrometric…

Aquigen Bio Strengthens Pharmaceutical Research with High-Purity Icatibant Impur …
Aquigen Bio, a trusted supplier of pharmaceutical reference standards, today announced the expansion of its Icatibant Impurity Standards portfolio, designed to support drug developers, analytical laboratories, and research organizations with reliable materials for impurity profiling and quality control.
Icatibant, a selective bradykinin B2 receptor antagonist, is widely used in the treatment of hereditary angioedema (HAE). Given its peptide-based structure, Icatibant is prone to the formation of impurities during synthesis and storage.…
More Releases for Butamirate
Global Cough Suppressant Industry | Innovative Delivery Systems Reshape Market | …
The Global Cough Suppressant Drugs Market is expected to grow at a CAGR of 3.4% during the forecast period (2024-2031).
The Cough Suppressant Drugs Market receives exhaustive analysis from DataM Intelligence, delivering stakeholders essential market data, emerging industry patterns, and strategic business intelligence. This in-depth research explores the competitive landscape in detail, evaluating market leaders across multiple dimensions including their innovative product offerings, competitive pricing strategies, financial performance metrics, strategic growth…
Cough Suppressant Drugs Market Presents a Strategic Forecast (2024-2034) on Mark …
Prophecy Market Insights serves as the compass guiding businesses through the dynamic landscape of the Cough Suppressant Drugs Market. In essence, it is the systematic process of gathering, analyzing, and interpreting information about this specific market, its consumers, and competitors. This invaluable tool empowers businesses operating in the Cough Suppressant Drugs Market to make informed decisions, mitigate risks, and seize opportunities. By unraveling the intricacies of consumer behavior, industry trends,…
Cough Suppressant Drugs Market- Trends, Analysis and Forecast till 2030
PMI's Latest Report, Cough Suppressant Drugs Market report is analyzed and studied on the basis of a comprehensive backdrop analysis. Hence, the Global Cough Suppressant Drugs Market report focuses on the information related to the several market segmentations, geographical segmentation, market dynamics, market growth factors, and a complete study of the competitive overview of this market. In addition, the Market research report covers a detailed study of company profiles, which…
The Global Cough Suppressant Drugs Market Size Was Valued At Us$ 1100.4 Mn In 20 …
A cough is a most common symptom of various upper and lower respiratory tract infections. It affects a large proportion of the population leading them to seek medical attention. According to the data published by Journal of the Association of Physicians of India in 2016, cough was identified as the sixth most common reason for hospital outpatient department visits. Cough suppressants, also known as antitussives, are medications that help to…
Cough Suppressant Drugs Market Estimated To Witness a Phenomenal Growth by 2026 …
A cough is a most common symptom of various upper and lower respiratory tract infections. It affects a large proportion of the population leading them to seek medical attention. According to the data published by Journal of the Association of Physicians of India in 2016, cough was identified as the sixth most common reason for hospital outpatient department visits. Cough suppressants, also known as antitussives, are medications that help to…
Cough Suppressant Drugs Market Research Study including Growth Factors, Types an …
A cough is a most common symptom of various upper and lower respiratory tract infections. It affects a large proportion of the population leading them to seek medical attention. According to the data published by Journal of the Association of Physicians of India in 2016, cough was identified as the sixth most common reason for hospital outpatient department visits. Cough suppressants, also known as antitussives, are medications that help to…