Press release
Companion Diagnostic Tests in Oncology Market Comprehensive Analysis of Growth Trends and Future Opportunities - Estimated to reach USD 17.0 billion by 2034
The companion diagnostic tests in oncology market is experiencing significant growth, driven by the increasing demand for personalized cancer treatments and the integration of targeted therapies. Companion diagnostic tests are crucial tools in oncology that help identify biomarkers indicating a patient's suitability for a specific therapy. This enables more precise treatment plans, better therapeutic outcomes, and minimized adverse effects. These tests are primarily categorized by detection techniques - protein detection, DNA detection, and other methods - and biomarkers, such as EGFR, KRAS, HER2, BRAF V600E, and others.In 2023, the companion diagnostic tests in oncology market was valued at approximately US$ 6.1 billion. It is anticipated to experience robust growth, with a Compound Annual Growth Rate (CAGR) of 9.5% from 2024 to 2034, reaching an estimated US$ 17.0 billion by the end of 2034. This growth is driven by the rising demand for targeted therapies, advances in genomic research, and a greater focus on personalized medicine in oncology care. The increasing prevalence of cancer globally, along with the need for more specific diagnostic tools to support targeted treatments, further propels the demand for companion diagnostic tests.
Examine key highlights and takeaways from our Report in this sample - https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=4684
Competitive Landscape
The companion diagnostic tests market in oncology is highly competitive and driven by technological innovation, research collaborations, and strategic partnerships. A mix of large pharmaceutical companies, diagnostic equipment manufacturers, and biotechnology firms contribute to a diverse competitive landscape. Key companies in the market include:
Abbott - Known for offering a broad portfolio of diagnostic tests and technologies, Abbott continues to advance in developing companion diagnostic tests for various cancer types and targeted therapies.
F. Hoffmann-La Roche AG - Roche stands out for its commitment to precision oncology, with a robust portfolio of diagnostic tests, particularly in DNA detection technologies and targeted biomarkers.
Genomic Health, Inc. - A pioneer in genomic testing, Genomic Health focuses on personalized diagnostic tests that align patients with therapies tailored to their specific genetic profiles.
QIAGEN, Agilent Technologies, Inc., and AGENDIA N.V. also maintain a strong presence, contributing technologies that detect biomarkers such as EGFR, BRAF V600E, KRAS, and HER2.
bioMérieux SA, Illumina, Inc., Siemens Healthineers, Thermo Fisher Scientific Inc., and BioGenex bring significant innovation to the detection methods and technologies, offering robust tools that integrate genomic research with clinical oncology workflows.
These companies focus on advancing molecular testing, next-generation sequencing, and bioinformatics tools to ensure high accuracy and efficiency in companion diagnostic tests. Collaborations, acquisitions, and investments in research and development are central strategies for maintaining leadership in this competitive market.
Emerging Trends
Several emerging trends are shaping the companion diagnostic tests market in oncology, ensuring advancements in technology and improved patient care outcomes.
One of the prominent trends is the shift towards personalized cancer therapies. Healthcare providers are increasingly adopting companion diagnostic tests to match cancer treatments with the specific molecular profile of an individual's tumor, ensuring more effective and targeted treatment regimens.
Another significant trend is the adoption of next-generation sequencing (NGS) technologies, which allows the detection of multiple genetic variations simultaneously. NGS is critical for identifying biomarkers like EGFR mutations, KRAS mutations, BRAF V600E, and others that play a role in targeted treatments.
There is also a growing demand for AI and machine learning integration, which enhances the accuracy of biomarker detection and data interpretation. AI algorithms support diagnostic tools by providing real-time data analysis, pattern recognition, and predictive insights, which improve treatment planning and patient outcomes.
Furthermore, regulatory approvals and standardizations in oncology diagnostics are becoming more stringent, ensuring that companion diagnostic tests meet high clinical and safety standards. This leads to a greater focus on stringent clinical trials and validation processes to comply with international healthcare regulations.
Market Dynamics
The companion diagnostic tests market in oncology is influenced by multiple factors, including technological advancements, regulatory policies, and clinical needs. A major driver for the market is the growing prevalence of cancer cases worldwide, which fuels the demand for early detection and accurate diagnostic tools. Companion diagnostic tests play a crucial role in ensuring effective treatment planning and improving patient outcomes by aligning therapies with the patient's unique genetic and molecular profile.
The market is also supported by government initiatives and healthcare investments in precision oncology. Healthcare systems across developed and developing regions are increasingly prioritizing personalized medicine, investing in advanced diagnostic tools, and supporting research initiatives to promote targeted cancer treatments.
However, the market faces challenges such as high costs of companion diagnostic tests and complex regulatory environments. Companion diagnostic technologies often require extensive validation, clinical trials, and compliance with stringent healthcare regulations, which can result in high development costs. Furthermore, competition among diagnostic companies to provide cost-effective solutions puts pressure on profit margins.
Visit our report to explore critical insights and analysis - https://www.transparencymarketresearch.com/companion-diagnostic-tests-market.html
Opportunities and Challenges
The companion diagnostic tests market presents significant opportunities driven by the growing need for targeted therapies and personalized oncology care. Innovations in molecular biology and genomics offer the development of more accurate and efficient diagnostic tests, allowing healthcare providers to select optimal therapies for individual cancer patients.
Collaborative efforts between pharmaceutical companies, diagnostic manufacturers, and genomics research institutions present opportunities to develop companion diagnostic tests that are integrated directly into targeted treatment workflows, ensuring seamless clinical application.
However, the market also faces challenges. The high cost of research and development, coupled with stringent clinical validation requirements, can be a barrier to the commercialization of advanced diagnostic technologies. Moreover, ensuring widespread accessibility of companion diagnostic tests in developing regions remains a significant challenge due to infrastructure limitations and budget constraints.
Future Outlook
The future of the companion diagnostic tests market in oncology looks highly promising, with an expected CAGR of 9.5% from 2024 to 2034. As technology continues to evolve, molecular diagnostics, artificial intelligence, and bioinformatics integration will drive innovations in companion diagnostic tests, offering faster, more accurate, and cost-effective solutions for early cancer detection and treatment planning.
The development of advanced liquid biopsy technologies and high-throughput sequencing methods will expand the market significantly. Liquid biopsy, in particular, is gaining traction as a non-invasive method for detecting cancer biomarkers, which reduces surgical risks and provides quick, accurate insights into a patient's cancer status.
Governments and healthcare organizations are anticipated to continue investing in personalized cancer research and infrastructure development, especially in emerging markets across Asia-Pacific, Latin America, and the Middle East. These regions present opportunities for companion diagnostic technologies that are both affordable and accessible.
Consumer Behavior
Oncology specialists, hospitals, research institutions, and pharmaceutical companies remain the primary consumers of companion diagnostic tests. Healthcare providers prioritize technological efficiency, accuracy, and clinical validation when choosing diagnostic tools. They prefer tests that provide quick, reliable biomarker detection, integrate seamlessly with surgical and imaging technologies, and comply with stringent clinical standards.
Pharmaceutical companies also leverage companion diagnostic tools to align clinical trials with targeted therapies, ensuring that patient selection for experimental treatments is based on accurate biomarker profiles.
Patients are increasingly aware of companion diagnostic tests, and there is a growing demand for personalized treatments that minimize side effects while maximizing therapeutic effectiveness. Healthcare consumers expect fast, non-invasive detection methods that reduce discomfort and provide actionable insights.
Regional Analysis
In North America, robust research initiatives, high healthcare investments, and technological advancements position the market as a leader in companion diagnostics. Regulatory frameworks and clinical validation requirements ensure that all diagnostic technologies meet stringent safety and effectiveness standards.
In Europe, the focus is on high-quality clinical research and development, with regulatory bodies like the European Medicines Agency setting strict guidelines. Europe's emphasis on precision oncology and targeted therapies drives the demand for accurate companion diagnostic tools.
In Asia-Pacific, there is a growing emphasis on precision oncology infrastructure, supported by increasing investments in healthcare technologies, research collaborations, and rising cancer prevalence. Emerging markets in China and India offer significant opportunities due to large patient populations and substantial investments in healthcare infrastructure.
Latin America and the Middle East are also witnessing growth, with increasing government initiatives focused on personalized medicine, targeted therapies, and healthcare infrastructure upgrades.
More Trending Reports by Transparency Market Research -
Blood Pressure Cuffs Market - https://www.transparencymarketresearch.com/blood-pressure-cuffs-market.html
Vaginal Slings Market - https://www.transparencymarketresearch.com/vaginal-slings-market.html
Stretcher Chairs Market - https://www.transparencymarketresearch.com/stretcher-chairs-market-2018-2026.html
Plasma Fractionation Market - https://www.transparencymarketresearch.com/plasma-fractionation-market.html
Biomarkers Market - https://www.transparencymarketresearch.com/biomarkers-market.html
Endoscopic Stricture Management Device Market - https://www.transparencymarketresearch.com/endoscopic-stricture-management-device-market.html
Flowable Hemostats Market - https://www.transparencymarketresearch.com/flowable-hemostats-market.html
Hormonal Contraceptive Market - https://www.transparencymarketresearch.com/hormonal-contraceptives-market.html
About Transparency Market Research
Transparency Market Research, a global market research company registered at Wilmington, Delaware, United States, provides custom research and consulting services. Our exclusive blend of quantitative forecasting and trends analysis provides forward-looking insights for thousands of decision makers. Our experienced team of Analysts, Researchers, and Consultants use proprietary data sources and various tools & techniques to gather and analyses information.
Our data repository is continuously updated and revised by a team of research experts, so that it always reflects the latest trends and information. With a broad research and analysis capability, Transparency Market Research employs rigorous primary and secondary research techniques in developing distinctive data sets and research material for business reports.
Contact:
Transparency Market Research Inc.
CORPORATE HEADQUARTER DOWNTOWN,
1000 N. West Street,
Suite 1200, Wilmington, Delaware 19801 USA
Tel: +1-518-618-1030
USA - Canada Toll Free: 866-552-3453
Website: https://www.transparencymarketresearch.com
Email: sales@transparencymarketresearch.com
Follow Us: LinkedIn| Twitter| Blog | YouTube
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Companion Diagnostic Tests in Oncology Market Comprehensive Analysis of Growth Trends and Future Opportunities - Estimated to reach USD 17.0 billion by 2034 here
News-ID: 3785416 • Views: …
More Releases from Transparency Market Research

Medium Voltage Fuse Market Outlook 2031: Global Market to Reach US$ 1.8 Billion …
The global medium voltage fuse market is steadily transitioning from a traditional grid-protection niche to a strategic enabler of modern power systems. Rising investments in renewable energy integration, large-scale electrification programs, and infrastructure upgrades are reshaping demand patterns worldwide. Medium voltage fuses-typically rated between 1 kV and 35 kV-are no longer viewed as passive safety components; instead, they are increasingly recognized as critical assets for grid stability, asset protection, and…
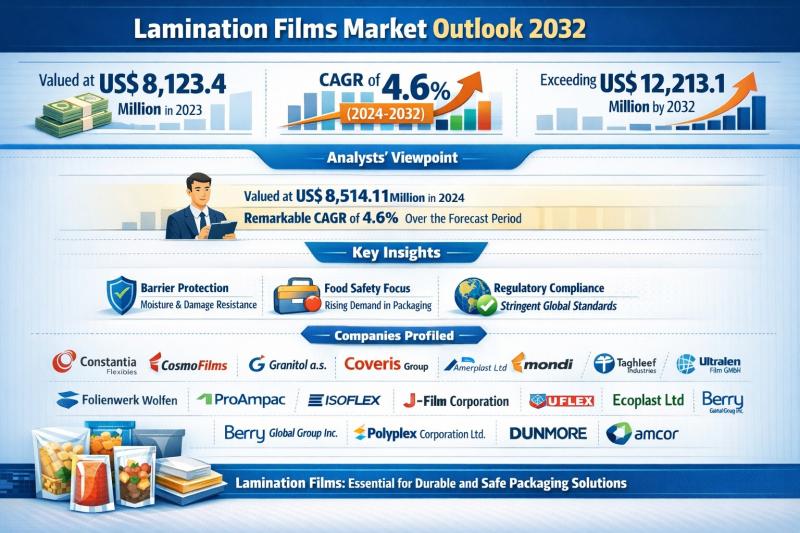
Lamination Films Market Outlook 2032: Global Industry Size to Surpass US$ 12.21 …
The global lamination films market was valued at US$ 8,123.4 million in 2023 and is forecast to reach US$ 8,514.1 million in 2024. Over the forecast period from 2024 to 2032, the market is projected to expand at a compound annual growth rate (CAGR) of 4.6%, ultimately exceeding US$ 12,213.1 million by 2032. This steady growth trajectory reflects the indispensable role of lamination films in modern packaging ecosystems across food,…

Global Curcumin Market Outlook 2031: Natural Antioxidant Demand, Regional Growth …
The global curcumin market is entering a structurally strong growth phase, underpinned by rising consumer preference for natural, plant-based ingredients and increasing clinical validation of curcumin's antioxidant and anti-inflammatory properties. As consumers shift away from synthetic additives and chemical-based therapeutics, curcumin is emerging as a high-value bioactive ingredient across , functional foods, cosmetics, and pharmaceuticals. Premiumization trends in organic products, combined with regulatory validation from food safety authorities, are expected…

Hemoglobin A1c Testing Devices Market to be Worth More Than USD 3.3 Bn by 2034 - …
The global Hemoglobin A1c (HbA1c) Testing Devices Market was valued at US$ 1.4 Bn in 2023 and is projected to expand at a CAGR of 7.7% from 2024 to 2034, reaching more than US$ 3.3 Bn by the end of 2034. The market growth is primarily attributed to the increasing global burden of diabetes, growing awareness about disease management, and technological advancements in diagnostic devices.
Get a concise overview of key…
More Releases for Companion
Companion Animal Medicine Market Healthy Companions, Healthy Profits: Trends in …
Companion Animal Medicine Market to reach over USD 41.71 billion by the year 2031 - Exclusive Report by InsightAce Analytic
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Companion Animal Medicine Market Size, Share & Trends Analysis Report By Animal Type (Dogs, Cats, And Horses), Product (Pharmaceuticals And Medicated Feed Additives), Distribution Channel (Veterinary Hospitals And Clinics), And Indication (Antibiotics and Parasiticides)- Market Outlook…
Companion Diagnostics Market - Companion Diagnostics Industry Size, Share, Analy …
Companion diagnostics are medical devices and tests that provide essential information for the safe and effective use of a corresponding drug or biological product. These diagnostics are critical in the field of personalized medicine, as they help identify patients who are most likely to benefit from a particular therapeutic product. The Global Companion Diagnostics Market was valued at US$ 5.59 Billion in 2022 and is projected to reach US$ 13.74…
Companion Animal Digital Technology Market
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Companion Animal Digital Technology Market By Type of Service (Telemedicine, Telehealth, Reminder Applications, Trackers, Wearables, and Platform Market Places) Trends, Industry Competition Analysis, COVID-19 Impact Analysis, Revenue and Forecast Till 2031." features detailed market analysis and an extensive study on the current trends, exploring its significant factors.
The Artificial Intelligence (AI) In Beauty and Cosmetics Market…
Companion Diagnostics Market - Transforming Patient Outcomes: Pioneering Compani …
Newark, New Castle, USA: The "Companion Diagnostics Market" provides a value chain analysis of revenue for the anticipated period from 2023 to 2031. The report will include a full and comprehensive analysis of the business operations of all market leaders in this industry, as well as their in-depth market research, historical market development, and information about their market competitors.
Companion Diagnostics Market: https://www.growthplusreports.com/report/companion-diagnostics-market/8673
This latest report researches the industry structure, sales, revenue,…
Companion Animal Vaccine – Improving The Health Of Companion Animals In A Cost …
The companion animal vaccines market is expected to witness significant growth during the forecast period due to launch of vaccines for companion animals. For instance, in December 2017, Zoetis Inc. announced the launch of Vanguard CIV H3N2/H3N8, the latest vaccine in the company’s canine influenza virus (CIV) portfolio. The new bivalent vaccine helps protect dogs against the two strains of the virus known to be circulating in the U.S.
Moreover, the…
Global Companion Diagnostics Market
The Global Companion Diagnostics (CDx) Market was valued at US$ 1,614.5 million in 2015 and is projected to expand at a CAGR of 12.0% during the forecast period (2016–2024), as highlighted in a new report published by Coherent Market Insights. Development of multiple biomarkers and targeted drug therapy is boosting research and development collaboration among the industry players. This is expected to improve the time to market for companion diagnostic (CDx) test…
