Press release
Liqomics Launches LymphoVista: Breakthrough in Minimal Residual Disease (MRD) Monitoring and Genotyping for Lymphoma Patients
Image: https://www.getnews.info/uploads/47d7fad53008388024cd78873530f9ca.pngLiqomics is proud to announce the launch of LymphoVista
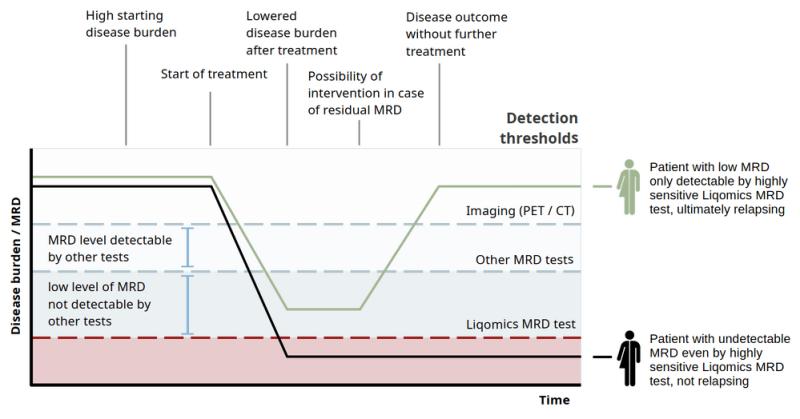
LymphoVista (https://liqomics.com/detect-lymphoma-lymphovista/) is an advanced ctDNA-based assay specifically designed for minimal residual disease (MRD) monitoring and genotyping in lymphoma patients. LymphoVista leverages high-sensitivity circulating tumor DNA (ctDNA) detection, setting a new standard in MRD testing across lymphomas including Hodgkin Lymphoma, Diffuse Large B-Cell Lymphoma (DLBCL), Central Nervous System (CNS) Lymphomas and Follicular Lymphoma.
Revolutionizing MRD Testing in Hodgkin Lymphoma and Other Lymphomas
LymphoVista addresses a critical need in genotyping and monitoring of MRD in lymphoma, providing a level of sensitivity, specificity, and accuracy that has been lacking in many current MRD assays. "Many existing cfDNA assays fail to meet the precision needed for lymphoma-specific MRD monitoring," said Sven Borchmann MD/PhD, founder and managing director of Liqomics. "With LymphoVista, we introduce a validated assay that can monitor MRD with accuracy as low as a few molecules of ctDNA per million enabling clinicians to make more data-driven treatment decisions."
LymphoVista HL Validation: High Sensitivity and Specificity
Validation studies confirm LymphoVista as a high-precision tool for MRD assessment. Key validation metrics include:
- Sensitivity > 91% and Specificity > 99.99% for variant detection (genotyping) allowing reliable identification of SNPs and InDels
- Limit of Detection (LoD) for MRD: 0.000654%, making it highly suitable for detecting minimal disease presence.
Clinical Validation Demonstrates Prognostic Power in Hodgkin Lymphoma
A clinical validation study for LymphoVista was performed in Hodgkin lymphoma (HL) involving 72 patients. This study demonstrated that MRD detection following two cycles of therapy effectively stratified patients based on risk. Patients MRD-negative patients after two cycles of chemotherapy had a 4-year progression-free survival (PFS) of 95.3% (95%-CI: 88.7 100) while patients who were MRD-positive after two cycles of chemotherapy had a 4-year PFS of 72.2% (95%-CI: 41.5-100) resulting in a hazard ratio of 6.9 (95%-CI: 4.5-10.6). In conclusion, MRD-negative patients showed very good outcomes, while MRD-positive patients had a significantly higher risk of relapse. This clinical performance study shows LymphoVista is a valuable tool for MRD detection in lymphoma, particularly Hodgkin lymphoma.
LymphoVista HL: Setting a New Benchmark for MRD-Guided Clinical Trials in Lymphoma
The launch of LymphoVista heralds a new era in personalized medicine for lymphoma, laying a foundation for MRD-guided clinical trials. The assay's validated accuracy and sensitivity support its use in clinical trials and can inform treatment decisions based on real-time MRD results.
For more information on LymphoVista or collaboration opportunities with Liqomics, please contact: https://liqomics.com/contact/
About Liqomics
Liqomics is a pioneering molecular diagnostics company focused on developing cutting-edge circulating tumor DNA (ctDNA) and cell-free DNA (cfDNA) assays to advance personalized medicine in oncology, with a strong emphasis on minimal residual disease (MRD) detection and genotyping in lymphomas and other hematological malignancies. Liqomics uses state-of-the-art technology for its assays which are rigorously validated to create highly sensitive diagnostic tools that enable precise disease monitoring and optimized treatment decisions.
The company is headquartered in Cologne, Germany. Liqomics' mission is to empower clinicians and patients with data-driven insights through validated cfDNA-based assays, ensuring that patients receive timely and effective care. Through its dedication to innovation and precision, Liqomics aims to set new standards in MRD monitoring and genotyping, enabling clinicians to make real-time adjustments in treatment plans based on the genetic profile and MRD status of each patient.
Media Contact
Company Name: Liqomics GmbH
Contact Person: Sven Borchmann MD/PhD Managing Director
Email: Send Email [http://www.universalpressrelease.com/?pr=liqomics-launches-lymphovista-breakthrough-in-minimal-residual-disease-mrd-monitoring-and-genotyping-for-lymphoma-patients]
Phone: (+49) 221 69 056 597
Address:Hermulheimer Str. 68
City: Koln 50969
Country: Germany
Website: https://liqomics.com/
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Liqomics Launches LymphoVista: Breakthrough in Minimal Residual Disease (MRD) Monitoring and Genotyping for Lymphoma Patients here
News-ID: 3740427 • Views: …
More Releases from Getnews

ICIC's Business Administration Program Equips Students for an AI Powered Future
Image: https://www.globalnewslines.com/uploads/2026/02/1772081606.jpg
As technology transforms construction and accelerates decision making, ICIC advances professional training to help managers lead with clarity in a data driven economy.
Delegacion Alvaro Obregon, CDMX - February 26, 2026 - As artificial intelligence reshapes industries and accelerates decision making across sectors, the Instituto de Capacitacion de la Industria de la Construccion (ICIC) continues to strengthen its academic portfolio with a program designed for modern leadership. ICIC's Business Administration…

RESET Hotel Launches Exclusive Stargazing Partnership with Celestron
Image: https://www.globalnewslines.com/uploads/2026/02/1772079550.jpg
A new desert hotel near Joshua Tree National Park unveils a first-of-its-kind stargazing partnership, elevating cosmic escapes this season.
Twentynine Palms, CA - February 26, 2026 - RESET Hotel [https://512857c0.streaklinks.com/CnmyKtFZ2enYBgVAhw75oHbJ/https%3A%2F%2Fwww.stayreset.com%2F], a new boutique hotel located 5 minutes from Joshua Tree National Park, has just announced a new partnership with Celestron, the leading manufacturer of telescopes and optical instruments, bringing an elevated stargazing experience to the California desert.
Image: https://i.postimg.cc/YShb8vLq/Celestron.jpg [https://postimages.org/]
As part…

iCAUR Makes Strategic Entry into the Middle East: Announcing V27's Global Launch …
Recently, Chery Group's new energy vehicle brand iCAUR's mid-to-large all-round hybrid SUV-the iCAUR V27-made its global launch in Dubai, UAE. This launch marks a key step in the brand's commitment to its Vision-"Co-create with users to build a warm, trustworthy new energy brand"-and serves as an important milestone for iCAUR, as "a premium new energy mobility brand driven by design and technology," in conveying its core proposition that "CLASSIC NEVER…

Veteran Combat Experience Meets Explosive Military SciFi
Image: https://www.aionewswire.com/storage/images/ckeditor//81J6D6xBOdL._SL1500__1772047334.jpg
ODAT: Orbital Drop Armor Team - Book 3: Imperial Storm
Retired U.S. Army officer LTC (R) Alexander R. Tambascia returns with the highly anticipated third installment of his military science fiction series, ODAT: Orbital Drop Armor Team - Book 3: Imperial Storm [https://www.amazon.com/gp/product/B0FPJ5R782?ref_=dbs_m_mng_rwt_calw_tkin_2&storeType=ebooks&qid=1772047291&sr=1-1]. Blending real-world combat experience with epic science fiction warfare, Imperial Storm delivers a gritty, authentic look at futuristic battlefields where survival is never guaranteed-and death may not…
More Releases for MRD
Minimal Residual Disease (MRD) Testing Market Demand in 2034
Rising adoption of next-generation sequencing (NGS), expanded use of MRD in hematologic malignancies, and growing integration of liquid biopsy technologies are propelling strong global market growth.
Download Full PDF Sample Copy of Market Report @
https://exactitudeconsultancy.com/request-sample/50110
Introduction
The Minimal Residual Disease (MRD) Testing Market is expanding rapidly as oncologists and diagnostic laboratories increasingly rely on highly sensitive molecular tests to detect trace levels of cancer cells that persist after treatment. MRD testing-using flow…
Lucintel Forecasts the Global MRD Testing Market to Reach $3.4 billion by 2031
According to a market report by Lucintel, the future of the global MRD testing market looks promising with opportunities in the hospitals and specialty clinic, diagnostic laboratory, and academic & research institutes markets. The global MRD testing market is expected to reach an estimated $3.4 billion by 2031 from $1.6 billion in 2024, at a CAGR of 11.7% from 2025 to 2031. The major drivers for this market are increasing…
MRD Testing Market Report: Trends, Forecast and Competitive Analysis to 2031
The future of the global MRD testing market looks promising with opportunities in the hospitals and specialty clinic, diagnostic laboratory, and academic & research institutes markets. The global MRD testing market is expected to reach an estimated $3.4 billion by 2031 with a CAGR of 11.7% from 2025 to 2031. The major drivers for this market are increasing occurrence of blood-related cancers, growing global elderly population, and progress in technology…
Lucintel Forecasts the Global MRD Testing Market to Reach $3.4 billion by 2031
According to a market report by Lucintel, the future of the global MRD testing market looks promising with opportunities in the hospitals and specialty clinic, diagnostic laboratory, and academic & research institutes markets. The global MRD testing market is expected to reach an estimated $3.4 billion by 2031 from $1.6 billion in 2024, at a CAGR of 11.7% from 2025 to 2031. The major drivers for this market are increasing…
Lucintel Forecasts the Global MRD Testing Market to Reach $3.4 billion by 2031
According to a market report by Lucintel, the future of the global MRD testing market looks promising with opportunities in the hospitals and specialty clinic, diagnostic laboratory, and academic & research institutes markets. The global MRD testing market is expected to reach an estimated $3.4 billion by 2031 from $1.6 billion in 2024, at a CAGR of 11.7% from 2025 to 2031.
According to a market report by Lucintel, the future…
Global MRD Testing Market Set to Reach USD 3.72 Billion by 2030
➤ Global Minimal Residual Disease (MRD) Testing Market to Reach USD 3.72 Billion by 2030, Driven by Technological Advancements and Rising Cancer Incidence
The global Minimal Residual Disease (MRD) Testing Market is projected to expand from USD 1.60 billion in 2023 to USD 3.72 billion by 2030, registering a robust Compound Annual Growth Rate (CAGR) of 12.8% during the forecast period.
➤ Competitive Landscape and Regional Demand
The MRD testing market…