Press release
Regulatory Affairs Outsourcing Market: Growth, Trends, and Key Insights
IntroductionThe regulatory affairs outsourcing market has emerged as a critical component in the global healthcare and pharmaceutical industries. Regulatory affairs, which involve ensuring that products meet the necessary legal and regulatory requirements to be marketed in different countries, have become more complex with the globalization of markets. As the healthcare and pharmaceutical sectors continue to expand, companies are increasingly outsourcing regulatory affairs to specialized service providers. This enables them to navigate complex regulatory landscapes, reduce operational costs, and focus on their core competencies. Regulatory affairs outsourcing includes services such as regulatory submissions, compliance management, labeling, and post-marketing surveillance, which are crucial for ensuring that pharmaceutical, biotechnology, medical devices, and other healthcare products meet regulatory standards.
Market Size
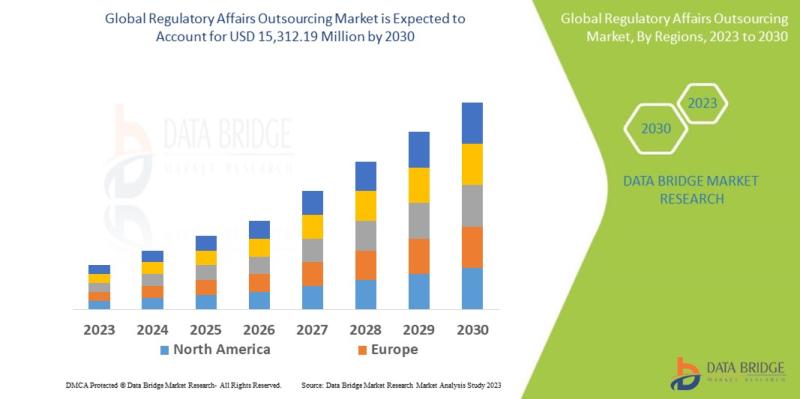
Data Bridge Market Research analyses that the regulatory affairs outsourcing market which was USD 9,389.20 million in 2022, would rocket up to USD 15,312.19 million by 2030, and is expected to undergo a CAGR of 10.66% during the forecast period. "Oncology" dominates the indication segment of the regulatory affairs outsourcing market owing to an increase in the prevalence of cancer. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
As healthcare and pharmaceutical companies increasingly operate on a global scale, the need for skilled regulatory affairs professionals to navigate the complex regulatory requirements in different regions continues to grow. Companies are opting to outsource these functions to mitigate the risks associated with non-compliance and ensure faster time-to-market for new products. The growing number of clinical trials, increasing pharmaceutical research and development (R&D) expenditure, and rising demand for biologics and medical devices also contribute to the market's expansion.
For More Information Visit https://www.databridgemarketresearch.com/reports/global-regulatory-affairs-outsourcing-market
Market Opportunities
The regulatory affairs outsourcing market presents several growth opportunities, particularly as the global regulatory environment becomes increasingly intricate. With the ongoing globalization of the pharmaceutical and healthcare industries, companies are seeking experienced service providers to help them navigate the ever-changing regulatory landscape. The growing complexity of regulatory guidelines, particularly for biologics, medical devices, and personalized medicine, is creating demand for more specialized outsourcing solutions.
One significant opportunity lies in the expanding market for regulatory affairs outsourcing in emerging economies, especially in regions like Asia-Pacific, Latin America, and the Middle East. These regions are experiencing rapid growth in healthcare, pharmaceutical manufacturing, and clinical research. With diverse regulatory frameworks, these markets require specialized regulatory support to ensure compliance with local regulations and international standards. Service providers who can offer region-specific regulatory expertise will be well-positioned to capitalize on these opportunities.
Another key opportunity is the increasing demand for digital solutions in regulatory affairs. The integration of digital technologies, such as artificial intelligence (AI), big data analytics, and machine learning, into regulatory processes is revolutionizing the way companies handle submissions, compliance monitoring, and post-market surveillance. Regulatory affairs outsourcing providers who can leverage these technologies to offer more efficient, data-driven solutions are likely to see increased demand.
Additionally, the increasing need for post-marketing surveillance services, as regulatory bodies worldwide focus more on patient safety and product monitoring, is driving opportunities in the outsourcing market. Regulatory affairs service providers that can handle not just pre-market approval but also ongoing regulatory compliance and risk management post-launch are in high demand.
Market Share
The regulatory affairs outsourcing market is highly fragmented, with a mix of large global outsourcing firms, mid-sized companies, and specialized niche service providers. Some of the key players in the market include Parexel International, Covance, Charles River Laboratories, IQVIA, and Medidata Solutions. These companies hold significant market share due to their established reputations, comprehensive service offerings, and global reach.
Additionally, there are numerous small and mid-sized firms specializing in regulatory affairs outsourcing, particularly in specific regions or niche markets. These smaller firms often offer specialized expertise in local regulations, enabling them to cater to clients in specific countries or markets with unique regulatory requirements.
The market share is also influenced by the level of specialization. Companies that provide end-to-end solutions, from regulatory submissions to post-market surveillance, capture a larger portion of the market as clients prefer to consolidate their outsourcing needs with a single service provider. On the other hand, smaller niche players that specialize in one area of regulatory affairs, such as labeling or clinical trials, often serve clients with specific needs.
Market Demand
The demand for regulatory affairs outsourcing services is closely tied to the pharmaceutical, biotechnology, medical device, and healthcare industries. The increasing pressure to comply with the complex and evolving regulatory environment is one of the major drivers of market demand. Regulatory requirements for drugs, biologics, medical devices, and even over-the-counter products have become more stringent and time-consuming, making it difficult for companies to manage these tasks in-house.
Pharmaceutical companies, in particular, are under pressure to expedite the approval process for new drugs while maintaining compliance with regulatory authorities such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and others. As a result, pharmaceutical companies are outsourcing regulatory affairs to service providers who can help them navigate these complex approval processes quickly and efficiently. The increasing cost of R&D and the need to reduce time-to-market further fuel the demand for regulatory outsourcing services.
The demand is also driven by the rising number of clinical trials being conducted globally. Clinical trials are a crucial part of the regulatory approval process, and regulatory affairs outsourcing plays a key role in ensuring that these trials meet regulatory standards across different regions. With an increasing number of clinical trials being conducted in emerging markets, the demand for local regulatory knowledge and expertise is rising.
Market Trends
The regulatory affairs outsourcing market is evolving, with several key trends shaping its future. One of the most notable trends is the growing adoption of digital solutions. The use of digital tools such as AI, machine learning, and big data analytics is transforming the way regulatory affairs are managed. These technologies help streamline regulatory submissions, track regulatory changes in real-time, and provide predictive analytics to improve decision-making. Outsourcing providers that can offer such advanced technological solutions are gaining a competitive edge.
Another significant trend is the increasing importance of regulatory affairs in the post-marketing phase. Regulatory authorities worldwide are putting more emphasis on post-market surveillance, pharmacovigilance, and ongoing compliance. This shift is driving the demand for outsourcing services that support companies in maintaining regulatory compliance after product launch. Regulatory affairs service providers offering post-marketing services, including adverse event reporting, safety monitoring, and product labeling, are seeing increased demand.
There is also a growing trend toward regional outsourcing. As healthcare and pharmaceutical companies expand into new markets, they require regulatory affairs expertise that is specific to local regulations. In response, outsourcing service providers are offering tailored solutions for different regions, particularly in emerging markets where regulatory frameworks can vary significantly from those in developed countries.
Furthermore, companies are increasingly seeking to outsource specific functions rather than full-service solutions. For example, a company may choose to outsource only the regulatory submission process or clinical trial management, rather than the entire regulatory affairs function. This trend is driven by the desire for flexibility, cost-effectiveness, and access to specialized expertise.
Market Growth
The regulatory affairs outsourcing market is poised for strong growth, fueled by the increasing complexity of global regulations and the need for efficient, cost-effective compliance. As pharmaceutical, biotechnology, and medical device companies face greater regulatory scrutiny, they are turning to specialized outsourcing partners to help navigate these challenges.
The market is also benefitting from the growth of emerging markets, where rising healthcare needs, increasing investments in the pharmaceutical industry, and the need for regulatory expertise create opportunities for outsourcing. The expansion of clinical trials in these regions further increases the demand for regulatory affairs services, providing a boost to the market.
Technological advancements are expected to play a major role in driving market growth. The increasing use of AI, automation, and digital solutions in regulatory affairs will make it easier for outsourcing providers to streamline processes, improve accuracy, and reduce costs. This, in turn, will drive more companies to outsource regulatory functions to third-party service providers.
Browse Trending Reports :
https://newsresmarket.blogspot.com/2024/11/cloud-storage-market-size-share-trends.html
https://newsresmarket.blogspot.com/2024/11/security-fencing-market-size-share.html
https://newsresmarket.blogspot.com/2024/11/industrial-plastic-market-size-share.html
https://newsresmarket.blogspot.com/2024/11/soft-drink-concentrates-market-size.html
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email: corporatesales@databridgemarketresearch.com
About Data Bridge Market Research:
Data Bridge set forth itself as an unconventional and neoteric Market research and consulting firm with unparalleled level of resilience and integrated approaches. We are determined to unearth the best market opportunities and foster efficient information for your business to thrive in the market. Data Bridge endeavors to provide appropriate solutions to the complex business challenges and initiates an effortless decision-making process.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Regulatory Affairs Outsourcing Market: Growth, Trends, and Key Insights here
News-ID: 3737774 • Views: …
More Releases from Data Bridge Market Research

Scented Candle Market Shows Strong Growth Driven by Wellness and Home Décor Tr …
The global scented candle market is on track for significant expansion, increasing from an estimated USD 3.60 billion in 2024 to USD 6.00 billion by 2032, registering a strong CAGR of 6.60%. Rising consumer interest in home ambiance, wellness, and premium lifestyle products continues to drive market demand.
Get More Detail: https://www.databridgemarketresearch.com/reports/global-scented-candle-market
Market Growth Drivers
The scented candle market has evolved beyond being just a decorative item. Key growth factors include:
Home Fragrance &…
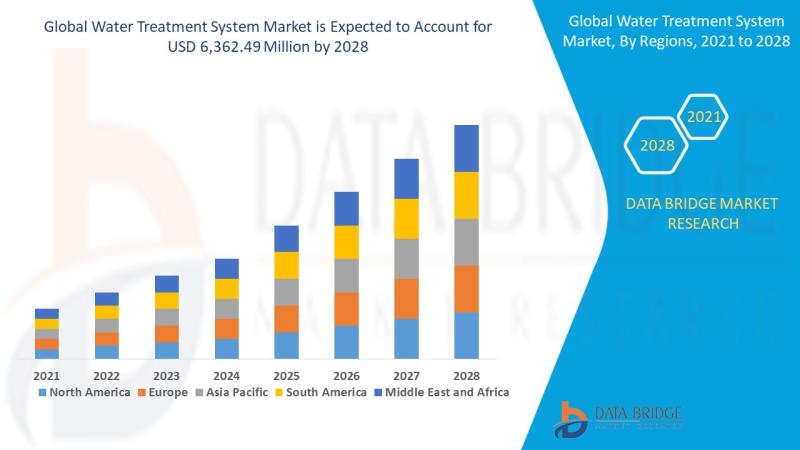
Water Treatment System Market: Sustaining the Future of Clean Water
Introduction
Understanding Water Treatment Systems
Water treatment systems are designed to purify and disinfect water for various uses-drinking, industrial processes, irrigation, and wastewater reuse. These systems eliminate contaminants such as bacteria, viruses, heavy metals, chemicals, and particulates, making water safe and sustainable for consumption and use.
Importance in Global Sustainability
Clean water is essential to life and industrial progress. With growing water demand and pollution, water treatment systems are now critical infrastructure across the…

Veterinary X-Ray Market Size, Analysis, Scope, Demand, Opportunities, Statistics
According to Data Bridge Market Research The global Veterinary X-Ray market size was valued at USD 915.19 million in 2024 and is projected to reach USD 1576.00 million by 2032, with a CAGR of 7.03 % during the forecast period of 2025 to 2032.
With increasing globalization and digital disruption, the Equine X-Ray Solutions Market is expanding across multiple industries, . Market research data indicates that businesses in the Companion Animal…

Veterinary X-Ray Market Size, Analysis, Scope, Demand, Opportunities, Statistics
According to Data Bridge Market Research The global Veterinary X-Ray market size was valued at USD 915.19 million in 2024 and is projected to reach USD 1576.00 million by 2032, with a CAGR of 7.03 % during the forecast period of 2025 to 2032.
With increasing globalization and digital disruption, the Equine X-Ray Solutions Market is expanding across multiple industries, . Market research data indicates that businesses in the Companion Animal…
More Releases for Regulatory
Medical Device Regulatory Affairs Market Medical Device Regulatory Affairs Marke …
"Medical Device Regulatory Affairs Market" in terms of revenue was estimated to be worth $ 6.7 billion in 2024 and is poised to reach $ 18.3 billion by 2034, growing at a CAGR of 10.8% from 2025 to 2034 according to a new report by InsightAce Analytic.
Request For Free Sample Pages:
https://www.insightaceanalytic.com/request-sample/1913
Latest Drivers Restraint and Opportunities Market Snapshot:
Key factors influencing the global medical device regulatory…
Medical Device & IVD Regulatory Affairs Outsourcing Market: Navigating Regulator …
Global healthcare landscape, the Medical Device & IVD Regulatory Affairs Outsourcing Market has emerged as a critical component ensuring the safe and compliant introduction of medical devices and in-vitro diagnostic products to the market. As the industry witnesses significant shifts and challenges, here's an in-depth analysis of the current trends, dynamics, and future prospects within this market segment.
Download sample PDF copy of report: https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=79264&utm_source=OpenPR_Ajay&utm_medium=OpenPR
Impact of COVID-19 on European Regulations
The outbreak of…
Regulatory Writing Market - Clear, Concise, Compliant: Redefining Regulatory Wri …
Newark, New Castle, USA - new report, titled Regulatory Writing Market The report has been put together using primary and secondary research methodologies, which offer an accurate and precise understanding of the Regulatory Writing market. Analysts have used a top-down and bottom-up approach to evaluate the segments and provide a fair assessment of their impact on the global Regulatory Writing market. The report offers an overview of the market, which…
Complex Regulatory Frameworks
It is challenging for new entrants to enter the FinTech industry because of its complex regulatory framework. All FinTech companies must comply with compliance requirements even before they begin operations, which increases their costs and creates a significant barrier for startups. While regulations are needed to protect consumers, a number of existing laws are slowing down the growth of many Indian FinTech companies, thereby extending their time to reach the…
South Africa Upstream Fiscal and Regulatory Report 2017 - Pending Legislation Cr …
Presented report, South Africa Upstream Fiscal and Regulatory Report 2017 - Pending Legislation Creates Regulatory Uncertainty, presents the essential information relating to the terms which govern investment into South Africa’s upstream oil and gas sector. The report sets out in detail the contractual framework under which firms must operate in the industry, clearly defining factors affecting profitability and quantifying the state’s take from hydrocarbon production. Considering political, economic and industry…
Regulatory Affairs Outsourcing Market (Services - Regulatory Submissions, Clinic …
This research study analyzes the market for regulatory affairs outsourcing services in terms of revenue (US$ Mn). The stakeholders of this report comprises the clinical research organizations. The global regulatory affairs outsourcing market has been broadly segmented on the basis of services (Regulatory Submissions, Clinical Trial Applications and Product Registrations, Regulatory Writing and Publishing, Regulatory Consulting and Legal Representation and others regulatory affairs, and Geography (North America, Europe, Asia Pacific,…