Press release
Advanced Therapy Medicinal Products Market Innovations in Patient Monitoring - Predicted to grow to USD 245.1 billion by 2034
The Advanced Therapy Medicinal Products (ATMP) market is rapidly evolving, driven by technological advancements and increasing demand for innovative therapies. These products, which encompass cell therapy, gene therapy, and tissue engineering, are poised to revolutionize the treatment landscape across various medical fields. This blog post provides an in-depth analysis of the ATMP market, exploring its growth outlook, competitive landscape, emerging trends, and future prospects from 2024 to 2034.The global Advanced Therapy Medicinal Products market was valued at approximately US$ 45.8 billion in 2023. This market is projected to experience substantial growth, advancing at a CAGR of 16.6% from 2024 to 2034, and is expected to surpass US$ 245.1 billion by the end of the forecast period. The surge in demand for ATMPs can be attributed to the increasing prevalence of chronic diseases, advancements in biotechnology, and the rising focus on personalized medicine. As healthcare systems evolve, there is a growing recognition of the potential of ATMPs to address previously untreatable conditions, thus driving investments and research in this innovative sector.
Discover essential conclusions and data from our Report in this sample -
https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=31298&utm_source=OpenPR_Ajay&utm_medium=OpenPR
Competitive Landscape
The ATMP market features a dynamic competitive landscape characterized by the presence of several prominent players dedicated to innovation and expansion. Companies such as Spark Therapeutics, Inc., Bluebird Bio, Inc., Novartis AG, UniQure N.V., MEDIPOST, Vericel Corporation, PHARMICELL Co., Ltd, and Organogenesis Inc. are leading the charge in developing and commercializing advanced therapies. These companies are heavily investing in research and development to enhance the efficacy and safety profiles of their products, focusing on diverse therapeutic applications, including oncology, neurology, and musculoskeletal disorders. Strategic collaborations, mergers, and acquisitions are common strategies employed by these organizations to expand their portfolios and reach new markets. For instance, partnerships with research institutions and academic centers are fostering innovation and facilitating the clinical development of novel therapies.
Emerging Trends
Several emerging trends are reshaping the ATMP market, primarily driven by advancements in research and technology. One significant trend is the increasing focus on personalized medicine, where treatments are tailored to individual patient profiles, enhancing efficacy and reducing adverse effects. This approach is particularly evident in gene therapy, where specific genetic mutations can be targeted to treat genetic disorders and certain types of cancers. Additionally, the rise of cell therapy is noteworthy, particularly in regenerative medicine, where stem cells are utilized to repair or replace damaged tissues. Furthermore, tissue engineering is gaining traction, with innovations in scaffold materials and biocompatible substances enabling the creation of functional tissues for transplant and repair. The demand for combination therapies, where ATMPs are used alongside traditional treatments, is also increasing, providing new avenues for comprehensive patient care.
Market Dynamics
The ATMP market is influenced by various dynamics that shape its growth trajectory. Key drivers include the increasing incidence of chronic and genetic diseases, advancements in biomanufacturing processes, and growing investments in research and development. The demand for innovative therapies that offer improved outcomes compared to conventional treatments is propelling market growth. However, challenges remain, such as regulatory hurdles and the high costs associated with developing and manufacturing ATMPs. Additionally, the complexity of these products often necessitates specialized manufacturing facilities, which can pose logistical challenges for companies. Varying reimbursement policies across different regions may also impact market penetration, particularly in developing countries where healthcare budgets are constrained.
Opportunities and Challenges
The ATMP market presents significant opportunities, particularly in emerging regions where healthcare infrastructure is improving and the demand for advanced therapies is rising. Regions such as Asia-Pacific and Latin America are witnessing an increase in healthcare investments, creating a favorable environment for ATMP adoption. Additionally, advancements in gene editing technologies, such as CRISPR-Cas9, are opening new avenues for developing targeted therapies. However, challenges persist, including the need for ongoing innovation to meet the expectations of patients and healthcare providers. Ensuring patient safety and achieving positive clinical outcomes will be crucial in maintaining market confidence and fostering further growth. Furthermore, the complexity of ATMPs necessitates rigorous testing and validation processes, which can extend development timelines and increase costs.
Future Outlook
The future outlook for the Advanced Therapy Medicinal Products market is highly promising, with substantial growth anticipated through 2034. As technological advancements continue to unfold, we can expect to see further innovations in ATMPs that will enhance their efficacy and expand their applications. The increasing focus on personalized medicine and the growing acceptance of gene and cell therapies will drive demand for these innovative products. Moreover, the ongoing development of biomanufacturing techniques and regulatory frameworks is expected to facilitate faster market entry for new therapies. As the healthcare landscape evolves, the integration of ATMPs into standard treatment protocols will likely become more prevalent, transforming patient care across various medical fields.
Review our report to gain deeper insights and understanding -
https://www.transparencymarketresearch.com/advanced-therapy-medicinal-products-market.html
Consumer Behavior
Consumer behavior in the healthcare sector is shifting towards innovative and personalized treatment options that promise better outcomes and fewer side effects. Patients are increasingly informed about their health conditions and treatment alternatives, leading to a greater demand for advanced therapies that address their unique medical needs. The growing acceptance of gene and cell therapies, particularly in oncology and rare genetic disorders, reflects this trend. Patients are seeking treatments that not only manage symptoms but also address the underlying causes of their conditions, driving interest in ATMPs that offer potential cures. Moreover, the rise of telehealth services and increased accessibility to information are empowering patients to engage more actively in their healthcare decisions, further influencing their preferences for advanced therapeutic options.
Regional Analysis
Regionally, the Advanced Therapy Medicinal Products market demonstrates varying characteristics. North America is currently the largest market for ATMPs, driven by advanced healthcare infrastructure, high levels of research funding, and a favorable regulatory environment that supports innovation. The European market is also experiencing significant growth, bolstered by strong support for research initiatives and increasing adoption of advanced therapies in clinical practice. In contrast, the Asia-Pacific region is expected to exhibit the fastest growth rate during the forecast period, fueled by rising healthcare investments, an expanding patient base, and increasing awareness of advanced therapeutic options. Emerging markets in Latin America and the Middle East also present substantial growth opportunities as governments invest in healthcare advancements and expand access to quality medical care.
Buy this Premium Research Report and unlock critical data and findings -
https://www.transparencymarketresearch.com/checkout.php?rep_id=31298<ype=S&utm_source=OpenPR_Ajay&utm_medium=OpenPR
More Trending Reports by Transparency Market Research -
Helicobacter Pylori (H. Pylori) Non-invasive Testing Market - https://www.transparencymarketresearch.com/helicobacter-pylori-non-invasive-testing-market.html
Surgical Staplers Market - https://www.transparencymarketresearch.com/surgical-staplers-market.html
HPLC Market - https://www.transparencymarketresearch.com/hplc-market.html
Spinal Implants Market - https://www.transparencymarketresearch.com/spinal-implants-market.html
About Transparency Market Research
Transparency Market Research, a global market research company registered at Wilmington, Delaware, United States, provides custom research and consulting services. Our exclusive blend of quantitative forecasting and trends analysis provides forward-looking insights for thousands of decision makers. Our experienced team of Analysts, Researchers, and Consultants use proprietary data sources and various tools & techniques to gather and analyses information.
Our data repository is continuously updated and revised by a team of research experts, so that it always reflects the latest trends and information. With a broad research and analysis capability, Transparency Market Research employs rigorous primary and secondary research techniques in developing distinctive data sets and research material for business reports.
Contact:
Transparency Market Research Inc.
CORPORATE HEADQUARTER DOWNTOWN,
1000 N. West Street,
Suite 1200, Wilmington, Delaware 19801 USA
Tel: +1-518-618-1030
USA - Canada Toll Free: 866-552-3453
Website: https://www.transparencymarketresearch.com
Email: sales@transparencymarketresearch.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Advanced Therapy Medicinal Products Market Innovations in Patient Monitoring - Predicted to grow to USD 245.1 billion by 2034 here
News-ID: 3710442 • Views: …
More Releases from Transparency Market Research
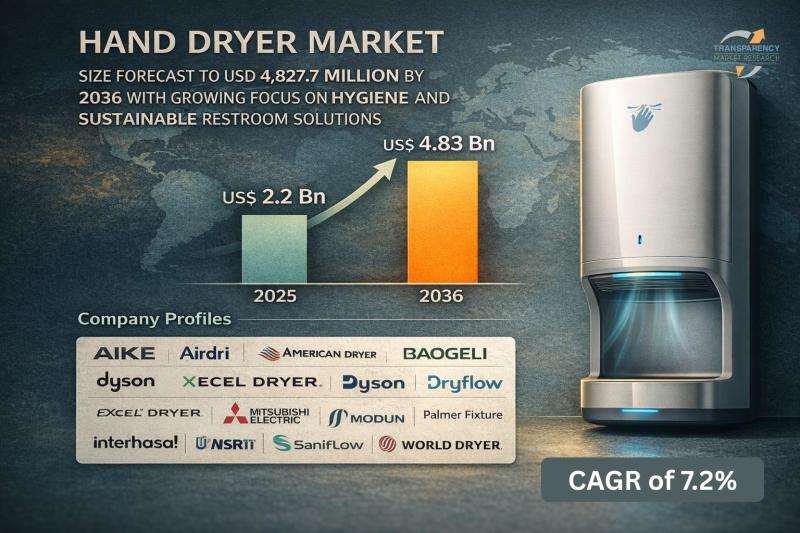
Hand Dryer Market Size Forecast to USD 4.83 Billion by 2036 with Growing Focus o …
Hand Dryer Market Outlook 2036
The global hand dryer market was valued at US$ 2.23 Billion in 2025 and is projected to reach US$ 4.83 Billion by 2036, expanding at a steady CAGR of 7.2% from 2026 to 2036. Market growth is driven by increasing emphasis on hygiene and sanitation, rising adoption in commercial infrastructure, and growing preference for eco-friendly and cost-effective hand drying solutions.
👉 Get sample market research report copy…
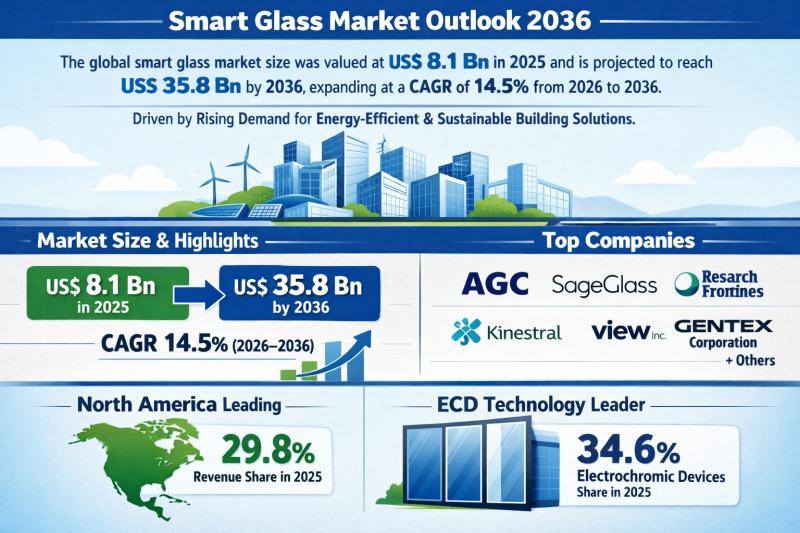
Smart Glass Market Outlook 2036: Projected to Reach USD 35.8 Billion at 14.5% CA …
The global smart glass market was valued at US$ 8.1 Bn in 2025 and is projected to surge to US$ 35.8 Bn by 2036, expanding at a robust CAGR of 14.5% from 2026 to 2036. This nearly 4.4x growth over eleven years underscores the accelerating demand for intelligent glazing solutions across commercial, residential, automotive, and infrastructure sectors.
North America emerged as the leading regional market in 2025, accounting for 29.8% of…
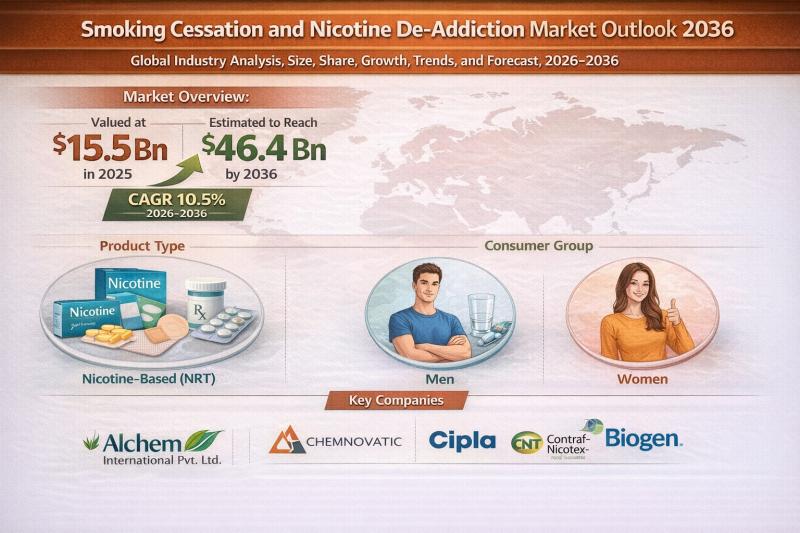
Smoking Cessation and Nicotine De-Addiction Market to Reach USD 46.4 Bn by 2036, …
The global smoking cessation and nicotine de-addiction market is witnessing strong and sustained growth, fueled by intensifying public health initiatives and rising awareness about the long-term consequences of tobacco use. Valued at USD 15.5 Bn in 2025, the market is projected to expand at a robust CAGR of 10.5% from 2026 to 2036, reaching USD 46.4 Bn by 2036.
Smoking cessation solutions encompass a wide range of products and services designed…

3D Imaging Market to be Worth USD 266 Bn by 2036 - By Component Type / By End-Us …
The global 3D imaging market is witnessing exponential growth, reflecting strong demand across healthcare, manufacturing, media, and industrial sectors. Valued at US$ 50 billion in 2025, the market is projected to reach US$ 266 billion by 2036, expanding at a robust CAGR of 18.2% from 2026 to 2036.
Get a concise overview of key insights from our Report in this sample -
https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=2743
This impressive trajectory highlights the rapid integration of advanced imaging…
More Releases for ATMP
Advanced Therapy Medicinal Products (ATMP) Market Is Going to Boom |• Novartis …
Worldwide Market Reports has recently released a comprehensive research study titled "Advanced Therapy Medicinal Products (ATMP) Market Size and Forecast 2026-2033: Breakdown by Manufacturers, Key Regions, Types, and Applications." The report has been developed using a combination of primary and secondary research methodologies, ensuring a detailed and accurate analysis. Drawing insights from both historical data and future projections, the study highlights the expected growth of the Advanced Therapy Medicinal Products…
ATMP Service Providers Market Driven by Rising Adoption of Personalized Medicine …
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global ATMP Service Providers Market by Type of Services (Analytics/CRO (Microbiology, Endotoxin, PCR, Flow Cytometry, ELISA, Container Closure, Sterilty, and Pre-clinical), Quality (Documentation, QMS And Regulations), CMO (HQ/GMP plasmid DNA, MSC Manufacture, Pluripotent Stem Cells), Logistics (Storage, Stability And Transport)), Application (GTMP(Gene Therapy Medicinal Products), sCTMP(somatic Cells Therapy Medicinal Products) and TEP(Tissue Engineered Products))-Trends, Industry Competition…
Lambda Biologics Secures Saxony Grant for ATMP Development with ORGANOIDSCIENCES
Image: https://www.getnews.info/uploads/88c7b29c2c2475f99f24c910a7580594.jpg
Lambda Biologics GmbH, located in the Leipzig Bio Cluster in Germany and led by CEO Andre Gerth, operates a biohealth testing platform. The company is growing as both a testing platform and a hub for knowledge and research, focusing not only on accurate test results but also on streamlining and advancing researchers' work processes.
Lambda Biologics received a public grant of the Free State of Saxony (Germany) in October to…
Advanced Therapy Medicinal Products (ATMP) Service Providers Market Exclusive Ov …
Advanced Therapy Medicinal Products (ATMP) Service Providers Market worth $34.59 Billion by 2030 - Exclusive Report by InsightAce Analytic
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global ATMP Service Providers Market by Type of Services (Analytics/CRO (Microbiology, Endotoxin, PCR, Flow Cytometry, ELISA, Container Closure, Sterilty, and Pre-clinical), Quality (Documentation, QMS And Regulations), CMO (HQ/GMP plasmid DNA, MSC Manufacture, Pluripotent Stem Cells), Logistics (Storage, Stability…
Advanced Therapy Medicinal Products (ATMP) Service Providers Market | Know the L …
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global ATMP Service Providers Market by Type of Services (Analytics/CRO (Microbiology, Endotoxin, PCR, Flow Cytometry, ELISA, Container Closure, Sterilty, and Pre-clinical), Quality (Documentation, QMS And Regulations), CMO (HQ/GMP plasmid DNA, MSC Manufacture, Pluripotent Stem Cells), Logistics (Storage, Stability And Transport)), Application (GTMP(Gene Therapy Medicinal Products), sCTMP(somatic Cells Therapy Medicinal Products) and TEP(Tissue Engineered Products))-Trends, Industry Competition…
ATMP Production Cost Analysis Report, Raw Materials Requirements, Costs and Key …
The latest report titled "ATMP Production Cost Report" by Procurement Resource, a global procurement research and consulting firm, provides an in-depth cost analysis ofa the production process of the ATMP. Read More: https://www.procurementresource.com/production-cost-report-store/atmp
Report Features - Details
Product Name - ATMP
Process Included - ATMP Production From Phosphorous Acid
Segments Covered
Manufacturing Process: Process Flow, Material Flow, Material Balance
Raw Material and Product/s Specifications: Raw Material Consumption, Product and Co-Product Generation, Capital Investment
Land and Site Cost:…