Press release
Regulatory Affairs Outsourcing Market Size, Share, Trends, Opportunities, Key Drivers and Growth Prospectus
Introduction:Regulatory affairs outsourcing involves delegating regulatory compliance tasks to third-party service providers. These tasks include obtaining and maintaining regulatory approvals for products, ensuring compliance with regulations, and managing regulatory documentation. Companies across various industries, particularly pharmaceuticals, biotechnology, medical devices, and food and beverages, outsource regulatory affairs to streamline operations, reduce costs, and ensure compliance with complex regulatory requirements. This article explores the regulatory affairs outsourcing market, focusing on market size, share, trends, growth, demand, and the factors driving its expansion.
Market Size:
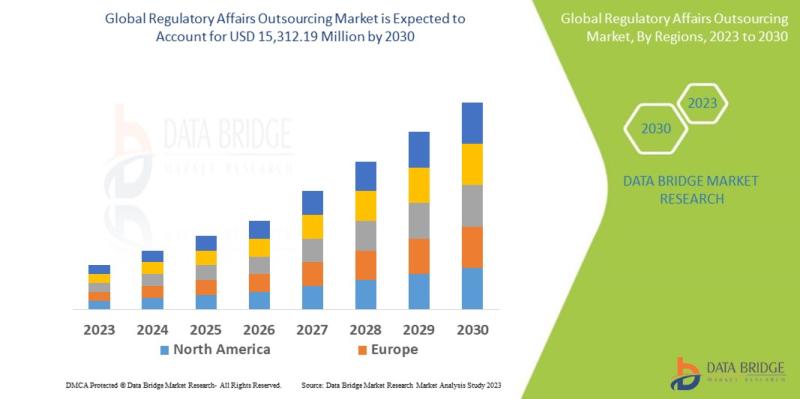
Data Bridge Market Research analyses that the regulatory affairs outsourcing market which was USD 9,389.20 million in 2022, would rocket up to USD 15,312.19 million by 2030, and is expected to undergo a CAGR of 10.66% during the forecast period. "Oncology" dominates the indication segment of the regulatory affairs outsourcing market owing to an increase in the prevalence of cancer. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
https://www.databridgemarketresearch.com/reports/global-regulatory-affairs-outsourcing-market
Some of the major players operating in the regulatory affairs outsourcing market are:
Accell Clinical Research, LLC (U.S.)
Genpact (U.S.)
CRITERIUM, INC. (U.S.)
PRA Health Sciences (U.S.)
Promedica International(U.S.)
WuXiAppTec (China)
Medpace (U.S.)
PPD Inc. (U.S.)
Charles River Laboratories (U.S.)
ICON plc (U.S.)
Covance (U.S.)
Parexel International Corporation. (U.S.)
Freyr, Navitas Clinical Research, Inc. (U.S.)
Medelis, Inc. (U.S.)
Sciformix (U.S.)
Tech Tammina(U.S.)
Acorn Regulatory Consultancy Services Ltd. (Ireland)
BIOMAPAS (Lithuania)
REGULATORY PROFESSIONALS (Australia)
CompareNetworks, Inc. (U.S.)
Market Share:
The regulatory affairs outsourcing market is highly competitive, with numerous players operating globally and regionally. Key players include ICON plc, PAREXEL International Corporation, Charles River Laboratories International, Inc., Covance Inc., and WuXi AppTec. ICON plc leads the market with its comprehensive regulatory services and extensive global presence. PAREXEL International Corporation is known for its specialized regulatory consulting and outsourcing solutions. Charles River Laboratories International, Inc., Covance Inc., and WuXi AppTec are also prominent players, offering a wide range of regulatory affairs services to clients across various industries.
Market Trends
Several key trends are shaping the regulatory affairs outsourcing market. One of the most significant trends is the increasing focus on regulatory compliance in emerging markets. As pharmaceutical and biotechnology companies expand their operations into emerging regions, they face complex regulatory environments that require specialized expertise. Outsourcing regulatory affairs to local service providers helps these companies navigate the regulatory landscape, ensuring compliance and facilitating market entry.
Another notable trend is the growing adoption of digital technologies and automation in regulatory affairs. The use of advanced software solutions, artificial intelligence (AI), and machine learning (ML) is transforming regulatory processes, enhancing efficiency, and reducing the risk of errors. Digital platforms enable real-time tracking of regulatory submissions, automated document management, and streamlined communication between stakeholders. This trend is driving the demand for technology-enabled regulatory outsourcing services.
The increasing focus on personalized medicine and advanced therapies is also influencing the market. The development and commercialization of personalized treatments and advanced therapies, such as gene and cell therapies, require specialized regulatory expertise. Companies are outsourcing regulatory affairs to service providers with experience in these areas, ensuring compliance with stringent regulatory requirements and accelerating the approval process.
Market Growth:
The regulatory affairs outsourcing market is poised for substantial growth in the coming years. Several factors contribute to this growth, including the rising costs of in-house regulatory operations. Maintaining an in-house regulatory affairs team can be expensive, particularly for small and mid-sized companies. Outsourcing regulatory tasks to specialized service providers allows companies to reduce costs, access specialized expertise, and focus on core business activities.
Technological advancements are also playing a crucial role in market growth. The adoption of digital technologies and automation is transforming regulatory affairs, enhancing efficiency, and reducing the risk of errors. Advanced software solutions, AI, and ML are streamlining regulatory processes, enabling real-time tracking of submissions, and improving document management. These advancements are driving the demand for technology-enabled regulatory outsourcing services.
The expanding pharmaceutical and biotechnology sectors are another significant factor contributing to market growth. The increasing development of new drugs, biologics, and medical devices is driving the demand for regulatory affairs services. Companies are outsourcing regulatory tasks to ensure compliance with regulatory requirements, facilitate market entry, and accelerate product approvals.
Market Demand:
The demand for regulatory affairs outsourcing is driven by several key factors. One of the primary drivers is the increasing complexity of regulatory frameworks. Regulatory requirements are becoming more stringent and complex, particularly in highly regulated industries such as pharmaceuticals and medical devices. Companies are outsourcing regulatory tasks to specialized service providers to ensure compliance with these complex requirements, avoid regulatory delays, and reduce the risk of non-compliance.
The growing need for specialized expertise is also driving market demand. Regulatory affairs require specialized knowledge and expertise, particularly for navigating complex regulatory environments and ensuring compliance with diverse regulations. Companies are outsourcing regulatory tasks to service providers with the necessary expertise and experience, enabling them to focus on core business activities and improve operational efficiency.
The rising costs of in-house regulatory operations are another significant factor driving demand. Maintaining an in-house regulatory affairs team can be expensive, particularly for small and mid-sized companies. Outsourcing regulatory tasks to specialized service providers allows companies to reduce costs, access specialized expertise, and focus on core business activities. This trend is particularly prominent in the pharmaceutical and biotechnology sectors, where regulatory compliance is critical to product development and commercialization.
Factors Driving Growth:
Several factors are driving the growth of the regulatory affairs outsourcing market. Firstly, the increasing complexity of regulatory frameworks is a major growth driver. Regulatory requirements are becoming more stringent and complex, particularly in highly regulated industries such as pharmaceuticals and medical devices. Companies are outsourcing regulatory tasks to specialized service providers to ensure compliance with these complex requirements, avoid regulatory delays, and reduce the risk of non-compliance.
Secondly, the rising costs of in-house regulatory operations are fueling market growth. Maintaining an in-house regulatory affairs team can be expensive, particularly for small and mid-sized companies. Outsourcing regulatory tasks to specialized service providers allows companies to reduce costs, access specialized expertise, and focus on core business activities. This trend is particularly prominent in the pharmaceutical and biotechnology sectors, where regulatory compliance is critical to product development and commercialization.
Technological advancements are also playing a crucial role in driving market growth. The adoption of digital technologies and automation is transforming regulatory affairs, enhancing efficiency, and reducing the risk of errors. Advanced software solutions, AI, and ML are streamlining regulatory processes, enabling real-time tracking of submissions, and improving document management. These advancements are driving the demand for technology-enabled regulatory outsourcing services.
The expanding pharmaceutical and biotechnology sectors are another significant factor contributing to market growth. The increasing development of new drugs, biologics, and medical devices is driving the demand for regulatory affairs services. Companies are outsourcing regulatory tasks to ensure compliance with regulatory requirements, facilitate market entry, and accelerate product approvals. The growing focus on personalized medicine and advanced therapies is further driving demand for specialized regulatory outsourcing services.
Conclusion:
The regulatory affairs outsourcing market is experiencing robust growth, driven by increasing complexity of regulatory frameworks, rising costs of in-house regulatory operations, and the growing need for specialized expertise. The market size and share are expanding, with key players leveraging innovative technologies and specialized expertise to maintain their competitive edge. Emerging trends such as the focus on regulatory compliance in emerging markets, adoption of digital technologies, and the growing emphasis on personalized medicine and advanced therapies are shaping the market landscape. The demand for regulatory affairs outsourcing is rising, fueled by the need for efficient and cost-effective regulatory solutions in highly regulated industries. As the market evolves, it is expected to witness further innovations and expanded applications, ensuring sustained growth in the years to come.
Browse Trending Reports:
https://marketresearchnewsdbmr.blogspot.com/2024/07/modified-wheat-starch-market-size-share_29.html
https://marketresearchnewsdbmr.blogspot.com/2024/07/automotive-parts-packaging-market-size.html
https://marketresearchnewsdbmr.blogspot.com/2024/07/laundry-detergent-market-size-share_29.html
https://marketresearchnewsdbmr.blogspot.com/2024/07/access-control-market-size-share-demand.html
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email: corporatesales@databridgemarketresearch.com
About Data Bridge Market Research:
Data Bridge set forth itself as an unconventional and neoteric Market research and consulting firm with unparalleled level of resilience and integrated approaches. We are determined to unearth the best market opportunities and foster efficient information for your business to thrive in the market. Data Bridge endeavors to provide appropriate solutions to the complex business challenges and initiates an effortless decision-making process.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Regulatory Affairs Outsourcing Market Size, Share, Trends, Opportunities, Key Drivers and Growth Prospectus here
News-ID: 3607183 • Views: …
More Releases from Data Bridge Market Research

Scented Candle Market Shows Strong Growth Driven by Wellness and Home Décor Tr …
The global scented candle market is on track for significant expansion, increasing from an estimated USD 3.60 billion in 2024 to USD 6.00 billion by 2032, registering a strong CAGR of 6.60%. Rising consumer interest in home ambiance, wellness, and premium lifestyle products continues to drive market demand.
Get More Detail: https://www.databridgemarketresearch.com/reports/global-scented-candle-market
Market Growth Drivers
The scented candle market has evolved beyond being just a decorative item. Key growth factors include:
Home Fragrance &…
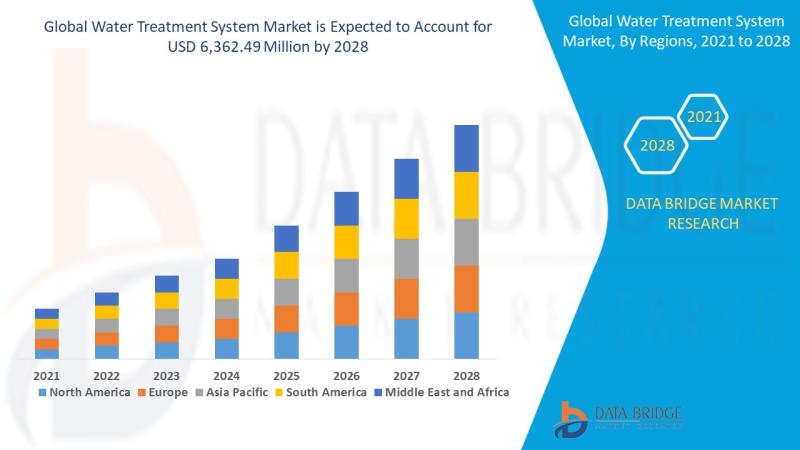
Water Treatment System Market: Sustaining the Future of Clean Water
Introduction
Understanding Water Treatment Systems
Water treatment systems are designed to purify and disinfect water for various uses-drinking, industrial processes, irrigation, and wastewater reuse. These systems eliminate contaminants such as bacteria, viruses, heavy metals, chemicals, and particulates, making water safe and sustainable for consumption and use.
Importance in Global Sustainability
Clean water is essential to life and industrial progress. With growing water demand and pollution, water treatment systems are now critical infrastructure across the…

Veterinary X-Ray Market Size, Analysis, Scope, Demand, Opportunities, Statistics
According to Data Bridge Market Research The global Veterinary X-Ray market size was valued at USD 915.19 million in 2024 and is projected to reach USD 1576.00 million by 2032, with a CAGR of 7.03 % during the forecast period of 2025 to 2032.
With increasing globalization and digital disruption, the Equine X-Ray Solutions Market is expanding across multiple industries, . Market research data indicates that businesses in the Companion Animal…

Veterinary X-Ray Market Size, Analysis, Scope, Demand, Opportunities, Statistics
According to Data Bridge Market Research The global Veterinary X-Ray market size was valued at USD 915.19 million in 2024 and is projected to reach USD 1576.00 million by 2032, with a CAGR of 7.03 % during the forecast period of 2025 to 2032.
With increasing globalization and digital disruption, the Equine X-Ray Solutions Market is expanding across multiple industries, . Market research data indicates that businesses in the Companion Animal…
More Releases for Regulatory
Medical Device Regulatory Affairs Market Medical Device Regulatory Affairs Marke …
"Medical Device Regulatory Affairs Market" in terms of revenue was estimated to be worth $ 6.7 billion in 2024 and is poised to reach $ 18.3 billion by 2034, growing at a CAGR of 10.8% from 2025 to 2034 according to a new report by InsightAce Analytic.
Request For Free Sample Pages:
https://www.insightaceanalytic.com/request-sample/1913
Latest Drivers Restraint and Opportunities Market Snapshot:
Key factors influencing the global medical device regulatory…
Medical Device & IVD Regulatory Affairs Outsourcing Market: Navigating Regulator …
Global healthcare landscape, the Medical Device & IVD Regulatory Affairs Outsourcing Market has emerged as a critical component ensuring the safe and compliant introduction of medical devices and in-vitro diagnostic products to the market. As the industry witnesses significant shifts and challenges, here's an in-depth analysis of the current trends, dynamics, and future prospects within this market segment.
Download sample PDF copy of report: https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=79264&utm_source=OpenPR_Ajay&utm_medium=OpenPR
Impact of COVID-19 on European Regulations
The outbreak of…
Regulatory Writing Market - Clear, Concise, Compliant: Redefining Regulatory Wri …
Newark, New Castle, USA - new report, titled Regulatory Writing Market The report has been put together using primary and secondary research methodologies, which offer an accurate and precise understanding of the Regulatory Writing market. Analysts have used a top-down and bottom-up approach to evaluate the segments and provide a fair assessment of their impact on the global Regulatory Writing market. The report offers an overview of the market, which…
Complex Regulatory Frameworks
It is challenging for new entrants to enter the FinTech industry because of its complex regulatory framework. All FinTech companies must comply with compliance requirements even before they begin operations, which increases their costs and creates a significant barrier for startups. While regulations are needed to protect consumers, a number of existing laws are slowing down the growth of many Indian FinTech companies, thereby extending their time to reach the…
South Africa Upstream Fiscal and Regulatory Report 2017 - Pending Legislation Cr …
Presented report, South Africa Upstream Fiscal and Regulatory Report 2017 - Pending Legislation Creates Regulatory Uncertainty, presents the essential information relating to the terms which govern investment into South Africa’s upstream oil and gas sector. The report sets out in detail the contractual framework under which firms must operate in the industry, clearly defining factors affecting profitability and quantifying the state’s take from hydrocarbon production. Considering political, economic and industry…
Regulatory Affairs Outsourcing Market (Services - Regulatory Submissions, Clinic …
This research study analyzes the market for regulatory affairs outsourcing services in terms of revenue (US$ Mn). The stakeholders of this report comprises the clinical research organizations. The global regulatory affairs outsourcing market has been broadly segmented on the basis of services (Regulatory Submissions, Clinical Trial Applications and Product Registrations, Regulatory Writing and Publishing, Regulatory Consulting and Legal Representation and others regulatory affairs, and Geography (North America, Europe, Asia Pacific,…