Press release
AI-Powered Solutions for Regulatory Excellence in the Medical Device Industry
In the fast-evolving landscape of the medical device industry, harnessing the power of Artificial Intelligence (AI) is not just advantageous but essential for maintaining regulatory excellence. At Celegence, we recognized the potential of AI to streamline complex processes and enhance efficiency in areas such as writing Clinical Evaluation Reports for Medical Devices.The Power of AI-Assisted Regulatory Writing
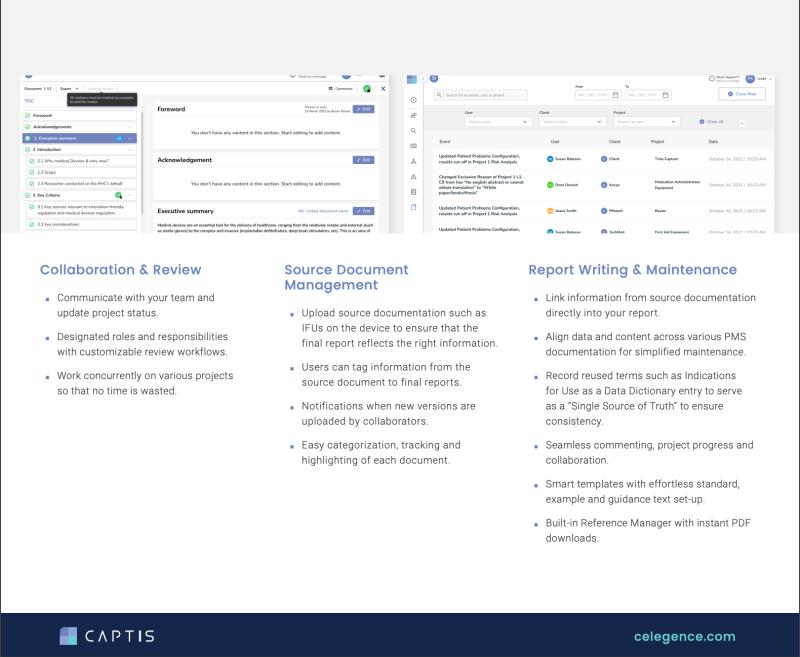
Our journey began with a clear objective: to optimize regulatory processes using AI while leveraging our regulatory expertise. By combining AI capabilities with our deep understanding of regulatory requirements, we developed prompt-based interactions that streamline data extraction during literature reviews for Clinical Evaluation Reports.
For instance, our AI-powered platform, CAPTIS, facilitates effortless extraction of essential data points such as study objectives, types, participant numbers, and follow-up durations.
"With contextual prompts tailored to specific requirements, writers can ensure consistency and accuracy in data presentation, thereby reducing manual errors and enhancing efficiency saving countless hours of manual review. As we harness the power of AI power, we elevate patient safety, accelerate drug approvals, and redefine healthcare communication," stated Sonia Veluchamy, CEO of Celegence.
AI Works Even Better with Instructions!
The beauty of AI is its ability to learn and adapt. The more specific your prompts, the better the results. For example, a writer can set up a prompt to automatically format participant age data in a consistent way across all reviewed studies. This eliminates the risk of inconsistencies and saves time and effort.
Merging AI and Regulatory Expertise
While AI excels at extraction, its true potential is unlocked when combined with deep regulatory knowledge. This synergy allows companies to navigate complex regulatory landscapes like Clinical Evaluations and Post-Market Clinical Follow-ups with greater agility and precision.
Here's a powerful example: imagine an AI tool that generates the initial framework for your State-of-the-Art Section in a clinical evaluation report. This AI assistant, guided by regulations like EU MDR and MEDDEV Rev 4, can provide information on clinical background, including epidemiology, treatment objectives, and the rationale for using your specific device. It can also give you information on the available alternative treatment options, and device-relevant safety and performance indicators. This foundational information can help medical writers overcome initial obstacles, and get straight to crafting a compelling narrative.
Celegence: Your Partner in AI-Powered Regulatory Excellence
At Celegence, we have extensive experience developing technology solutions for the medical device and pharma industry. We understand the challenges you face and the transformative power of automation and AI. Our team of experts combines cutting-edge technology with a deep understanding of regulatory frameworks.
We offer a range of solutions, from AI-powered platforms to niche regulatory software. Our goal is to empower companies to achieve excellence in their medical device or pharmaceutical regulatory pathways, paving the way for safer and more effective medical devices that ultimately improve patient outcomes worldwide.
Celegence LLC 1213 Butterfield Road, Downers Grove, IL 60515
Sonia Veluchamy is the CEO and co-founder of Celegence, a company dedicated to improving patient outcomes through intelligent regulatory compliance. With extensive regulatory expertise that spans all types of products, Celegence has gained rapid recognition, counting two top 5 pharmaceutical companies and several top 20 medical device companies among its esteemed clients. Through optimizing teams with tech-enabled services, Celegence supports these clients in navigating regulatory challenges, achieving compliance with evolving regulations, and reducing time to market in the most time and cost-efficient way.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release AI-Powered Solutions for Regulatory Excellence in the Medical Device Industry here
News-ID: 3440988 • Views: …
More Releases from Celegence
The Synergy Between Regulatory Intelligence and Pharmacovigilance
In the ever-evolving landscape of life sciences and healthcare, regulatory intelligence (RI) and pharmacovigilance (PV) play critical roles. Companies face multifaceted challenges that span across regulatory compliance, safety monitoring, and strategic decision-making. This is where experts like Celegence and Soterius come into play to bridge the gap between RI and PV.
Celegence offers a team of regulatory experts (https://www.celegence.com/services/regulatory-intelligence-solutions/) who understand the nuances of global regulations. They provide real-time…
More Releases for Sonia
Candle Market Detailed Analysis and Forecast to 2031 Fateh Industries, Sonia Sri …
[New York, December 2024] Candles have transcended their traditional role as mere sources of light to become integral elements in lifestyle products and home décor. With their diverse applications, from enhancing ambiance to serving as thoughtful gifts, the relevance of candles in contemporary society cannot be overstated. The candle market is characterized by an array of products including scented, decorative, and therapeutic candles, each catering to various customer preferences and…
Dynamic Speaker And Empowerment Coach Dr. Sonia L. Davidson Continues To Inspire …
Image: https://www.getnews.info/uploads/6f3f7794431f41dac1fa35472df5ab69.jpg
Fort Lauderdale, Florida - Dr. Sonia L. Davidson, an accomplished adult educator, speaker, and empowerment coach, is making a profound impact both nationally and internationally with her unwavering commitment to personal growth and helping others achieve their full potential. With over 25 years of experience in higher education, Dr. Davidson has become a beacon of inspiration for those striving to overcome adversity and succeed. Known for her dynamic presence,…
Cheil Appoints Sonia Grunbaum as Head of Business Development to Fuel Growth
Dallas, TX (October 8th, 2024) - Cheil has named Sonia Grunbaum as Head of Business Development to supercharge growth and expand the client roster. Known as one of the fastest-growing agencies in the U.S., this hire signals Cheil's ambition to accelerate growth even further.
In just five years, Cheil Dallas has grown to nearly 250 employees, delivering standout work for clients like Samsung Mobile. The agency specializes in connected experiences that…
Female Ready Made Clothes Market is Going to Boom | Sonia Rykiel, Valentino, Lac …
A Latest intelligence report published by AMA Research with title "Global Female Ready Made Clothes Market Outlook to 2028. A detailed study accumulated to offer Latest insights about acute features of the Female Ready Made Clothes market. This report provides a detailed overview of key factors in the Global Female Ready Made Clothes Market and factors such as driver, restraint, past and current trends, regulatory scenarios and technology development.
Definition:
Ready-made…
Female Ready Made Clothes Market Sets the Table for Continued Growth | Calvin Kl …
The latest study released on the Global Female Ready Made Clothes Market by AMA Research evaluates market size, trend, and forecast to 2027. The Female Ready Made Clothes market study covers significant research data and proofs to be a handy resource document for managers, analysts, industry experts and other key people to have ready-to-access and self-analyzed study to help understand market trends, growth drivers, opportunities and upcoming challenges and about…
Global Cream Foundation Market 2017 - SONIA RYKIEL, Benefit, Make Up for Ever, H …
Cream Foundation Market Research Report
A market study based on the " Cream Foundation Market " across the globe, recently added to the repository of Market Research, is titled ‘Global Cream Foundation Market 2017’. The research report analyses the historical as well as present performance of the worldwide Cream Foundation industry, and makes predictions on the future status of Cream Foundation market on the basis of this analysis.
Request For Sample…