Press release
Clinical Trial Management Systems Market is Expected to Grow at a CAGR of 11.50% to Reach US$ 1,840.40 Million by 2027
Clinical Trial Management Systems Market Report Overview :According to IMARC Group's latest report, titled "Clinical Trial Management Systems Market: Global Industry Trends, Share, Size, Growth, Opportunity and Forecast 2022-2027", offers a comprehensive analysis of the industry, which comprises insights on the market statistics. The report also includes competitor and regional analysis, and contemporary advancements in the global market.
A Closer Look at the Clinical Trial Management Systems Market Size | 2022
The global clinical trial management systems market reached a value of US$ 927.4 Million in 2021. Looking forward, IMARC Group expects the market to reach a value of US$ 1,840.40 Million by 2027 exhibiting a CAGR of 11.50% during 2022-2027.
Download a free sample report to get a detailed overview of the market: https://www.imarcgroup.com/clinical-trial-management-systems-market/requestsample
Clinical Trial Management Systems Market Insight :
A clinical trial management system (CTMS) represents a suite of tools that are utilized for effectively planning, tracking, and managing clinical trials in medical research studies. It primarily involves start-up timelines, reporting protocol information and accrual data, financial management, standardized billing processes and invoicing, subject visit tracking, etc. Moreover, CTMS standardizes clinical and operational data via advanced visualization and analytical techniques. Consequently, clinical trial management systems are extensively used by medical researchers to analyze, manage, and facilitate the transfer of patient data.
Clinical Trial Management Systems Market Current Trends & Future Predictions :
The escalating demand for personalized treatments and regenerative medicines is primarily driving the clinical trial management systems market. In line with this, the increasing usage of CTMS as a centralized and web-based enterprise resource to support complex clinical research studies in real-time is further catalyzing the market growth.
Additionally, the rising prevalence of new infectious and chronic diseases is propelling the adoption of advanced diagnostics and drug discovery tools, which, in turn, is acting as another significant growth-inducing factor.
Besides this, the inflating need for cloud-based CTMS to conduct trials of medical products, monitor the administration of vaccines, manage patient data, etc., on account of the sudden outbreak of the COVID-19 pandemic, is also augmenting the global market.
Furthermore, numerous technological advancements, including automated eClinical solutions for improving productivity, study reconciliation, data quality, etc., are anticipated to fuel the clinical trial management systems market over the forecasted period.
Inquire Before Buying : https://www.imarcgroup.com/request?type=report&id=4984&flag=F
Global Clinical Trial Management Systems Market 2022-2027 Analysis and Segmentation:
Competitive Landscape:
The competitive landscape of the market has been studied in the report with the detailed profiles of the key players operating in the market.
Advarra Inc., ArisGlobal LLC, BioClinica Inc., DataTrak International Inc., DZS Clinical Services Inc. (WDB Holdings Co. Ltd.), International Business Machines Corporation, Medidata Solutions Inc. (Dassault Systèmes SE), MedNet Solutions Inc., Oracle Corporation, Parexel International Corporation, RealTime Software Solutions LLC and Veeva Systems Inc.
The report has segmented the market on the basis of region, component, deployment mode and end user.
Breakup by Component:
• Software
• Services
Breakup by Deployment Mode:
• On-premises
• Cloud-based
Breakup by End User:
• Pharmaceuticals
• Contract Research Organization
• Others
Breakup by Region:
• North America: (United States, Canada)
• Asia Pacific: (China, Japan, India, South Korea, Australia, Indonesia, Others)
• Europe: (Germany, France, United Kingdom, Italy, Spain, Russia, Others)
• Latin America: (Brazil, Mexico, Others)
• Middle East and Africa
Ask Analyst for Customization and Explore Full Report with TOC & List of Figure: https://www.imarcgroup.com/clinical-trial-management-systems-market
If you want latest primary and secondary data (2022-2027) with Cost Module, Business Strategy, Distribution Channel, etc. Click request free sample report, published report will be delivered to you in PDF format via email within 24 to 48 hours of receiving full payment.
Key highlights of the report:
• Market Performance (2016-2021)
• Market Outlook (2022- 2027)
• Porter's Five Forces Analysis
• Market Drivers and Success Factors
• SWOT Analysis
• Value Chain
• Comprehensive Mapping of the Competitive Landscape
If you need specific information that is not currently within the scope of the report, we can provide it to you as a part of the customization.
IMARC Services Private Limited.
30 N Gould St Ste R
Sheridan, WY 82801 USA - Wyoming
Email: Sales@imarcgroup.com
Tel No:(D) +91 120 433 0800
Americas:- +1 631 791 1145 | Africa and Europe :- +44-702-409-7331 | Asia: +91-120-433-0800, +91-120-433-0800
IMARC Group is a leading market research company that offers management strategy and market research worldwide. We partner with clients in all sectors and regions to identify their highest-value opportunities, address their most critical challenges, and transform their businesses.
IMARC's information products include major market, scientific, economic and technological developments for business leaders in pharmaceutical, industrial, and high technology organizations. Market forecasts and industry analysis for biotechnology, advanced materials, pharmaceuticals, food and beverage, travel and tourism, nanotechnology and novel processing methods are at the top of the company's expertise.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Clinical Trial Management Systems Market is Expected to Grow at a CAGR of 11.50% to Reach US$ 1,840.40 Million by 2027 here
News-ID: 2892676 • Views: …
More Releases from IMARC Group
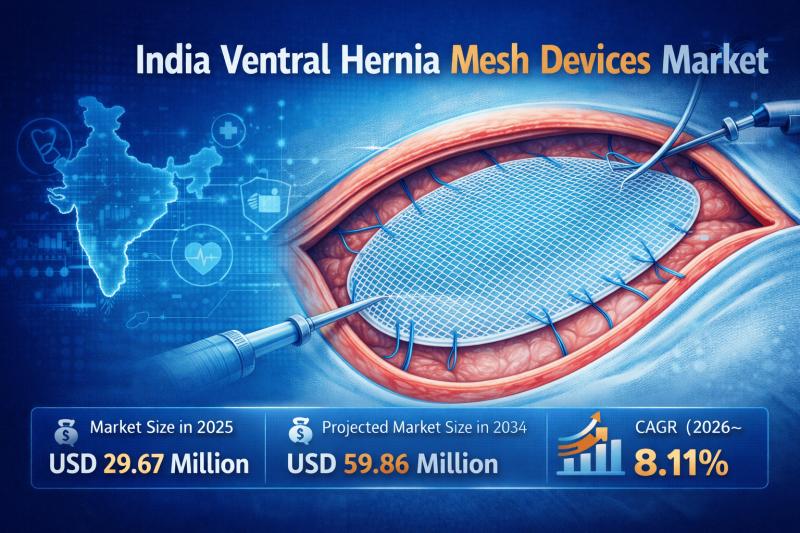
India Ventral Hernia Mesh Devices Market Expected to Reach USD 59.86 Million by …
IMARC Group's latest research publication, "India Ventral Hernia Mesh Devices Market Size, Share, Trends and Forecast by Mesh Type, Indication, Procedure, End Use, and Region, 2026-2034" the India ventral hernia mesh devices market size reached USD 29.67 Million in 2025. The market is expected to reach USD 59.86 Million by 2034, exhibiting a growth rate (CAGR) of 8.11% during 2026-2034.
Request a Sample Report: https://www.imarcgroup.com/india-ventral-hernia-mesh-devices-market/requestsample
What are Ventral Hernia Mesh Devices?
Ventral hernia…
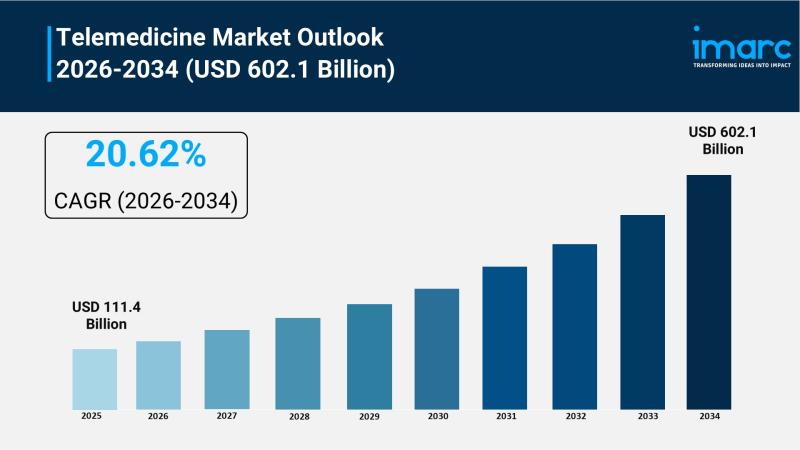
Telemedicine Market to Reach USD 602.1 Billion by 2034, Growing at a CAGR of 20. …
Market Overview:
The Telemedicine Market is experiencing rapid growth, driven by unprecedented integration of clinical AI assistants, progressive regulatory reimbursement frameworks, and the rapid maturation of the Internet of Medical Things (IoMT). According to IMARC Group's latest research publication, "Telemedicine Market : Global Industry Trends, Share, Size, Growth, Opportunity and Forecast 2025-2033", The global telemedicine market size was valued at USD 111.4 Billion in 2025. Looking forward, IMARC Group estimates the…
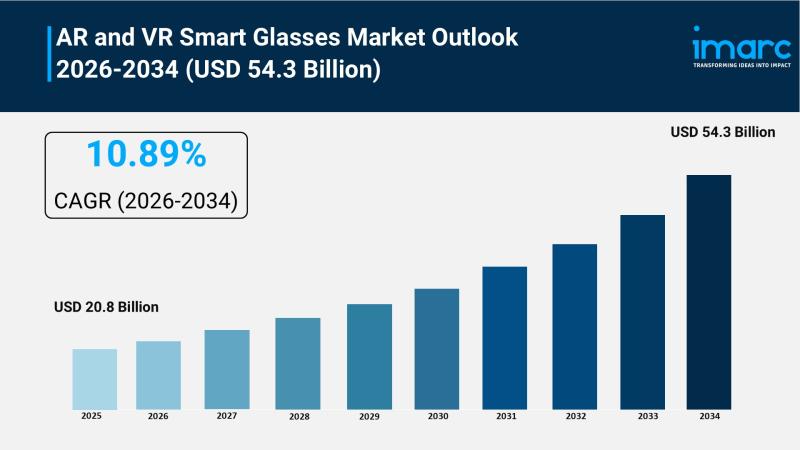
AR and VR Smart Glasses Market to Reach USD 54.3 Billion by 2034, Growing at a C …
Market Overview:
The AR and VR smart glasses market is experiencing rapid expansion, driven by breakthroughs in multimodal artificial intelligence, accelerated industrial digital twin integration, and the mass-market evolution of lifestyle-centric eyewear. According to IMARC Group's latest research publication, "AR and VR Smart Glasses Market: Global Industry Trends, Share, Size, Growth, Opportunity and Forecast 2025-2033", The global AR and VR smart glasses market size reached USD 20.8 Billion in 2025.…
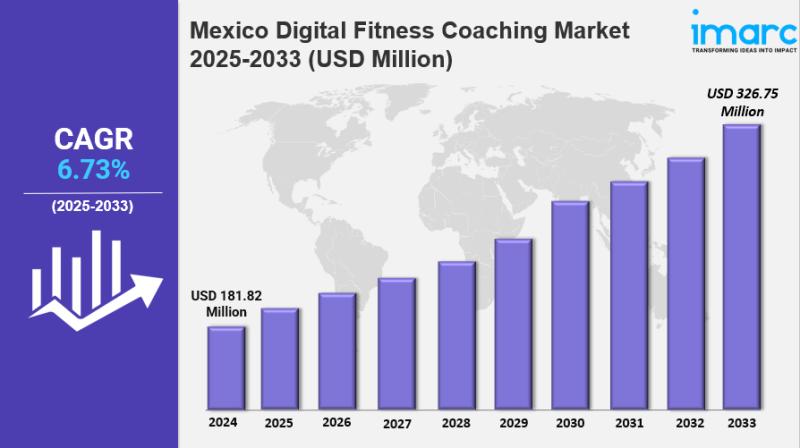
Mexico Digital Fitness Coaching Market Size, Growth, Key Players, Opportunity an …
IMARC Group has recently released a new research study titled "Mexico Digital Fitness Coaching Market Size, Share, Trends and Forecast by Streaming, Session, Revenue Model, Device, and Region, 2025-2033", offers a detailed analysis of the market drivers, segmentation, growth opportunities, trends and competitive landscape to understand the current and future market scenarios.
Mexico Digital Fitness Coaching Market Overview
The Mexico digital fitness coaching market size reached USD 181.82 Million in 2024. Looking…
More Releases for Clinical
Miami Clinical Research Sets the Standard for Clinical Trials
Miami Clinical Research, a frontrunner in the world of clinical trials and medical research, has emerged as the prime choice for global corporate pharmaceutical giants. With a deep understanding of the complexities of medical studies, the organization champions the crucial role of research in the evolution of transformative therapeutic interventions.
Miami, FL - Renowned as a first-rate center for professional medical exploration, Miami Clinical Research [https://miamiclinicalresearch.com] boasts state-of-the-art facilities, advanced technologies,…
E-Clinical Solutions Market: Revolutionizing Healthcare and Clinical Trials
Introduction
The e-Clinical solutions market has become a pivotal component of the healthcare and pharmaceutical industries. E-Clinical solutions refer to a set of software, tools, and platforms designed to streamline clinical trials and healthcare management. These solutions include electronic data capture (EDC), clinical trial management systems (CTMS), laboratory information management systems (LIMS), and other integrated tools that improve the efficiency, accuracy, and speed of clinical trials and healthcare services. The primary…
E-Clinical Solutions Market: Revolutionizing Clinical Trials
The e-clinical solutions market has experienced significant growth in recent years, driven by the increasing complexity of clinical trials and the need for efficient, accurate, and compliant data management. E-clinical solutions provide a comprehensive suite of tools and technologies to streamline clinical trial processes, accelerate drug development, and improve patient outcomes.
Market Size and Growth
The global e-clinical solutions market is estimated to be worth billions of dollars, with a significant portion…
Clinical Trials Management System Market Optimizing Clinical Trials: The Crucial …
Clinical Trials Management System Market to reach over USD 5.06 billion by the year 2031- Exclusive Report by InsightAce Analytic
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Clinical Trials Management System Market Size, Share & Trends Analysis Report By Solution Type (Enterprise and Site based), By Delivery Mode (Web & Cloud-based, On-premise), By Component (Software, Services), By End-user (Pharmaceutical and Biotechnology Firms, Medical…
Clinical Research and Clinical Trials Summit
Clinical Research 2019 has been designed in an interdisciplinary manner with a multitude of tracks to choose from every segment and provides you with a unique opportunity to meet up with peers from both industry and academia and establish a scientific network between them. We cordially invite all concerned people to come join us at our event and make it successful by your participation.
This is the premier interdisciplinary forum for…
E-Clinical Trial Solutions Market To Accelerating Clinical Development Technolog …
The study of the "Global e-Clinical Trial Solutions Market" provides the market size information and market trends along with the factors and parameters impacting it in both short and long term. The study ensures a 360° view, bringing out the complete key insights of the industry.
The Global e-Clinical Trial Solutions Market Research Report Forecast 2017-2021 is a valuable source of insightful data for business strategists. It provides the e-Clinical…
