Press release
The Growth Factors Market to look through AI-based genomics at a CAGR of 7.2%
The global Growth Factors Market is all set to witness a CAGR of 7.2% in the next decade. It is expected to reach US$ 3.6 Bn by the year 2031. With digitized communication channels making a beeline to the healthcare vertical, the advanced clinics are into virtual consultation mode, and this scenario is expected to continue even going forward. Regulatory approvals for this type of communication are on the way. Close to 70% of the public hospitals worldwide are being asked to adopt digital means. This would be the scene with the healthcare vertical in the subsequent period.The global growth factors market is expected to grow at a significant rate during the forecast period, owing to the increasing demand for growth factors in various applications, such as tissue engineering, drug development, and cell-based therapies. The market is also driven by the increasing demand for growth factors in the cosmetics industry, the growing research and development activities related to growth factors, and the increasing adoption of advanced technologies for the production of growth factors.
As per Persistence Market Research's latest revised industry analysis, the global growth factors market was valued at over US$ 1.7 Bn in 2020, and is expected to exhibit a CAGR of close to 7% over the forecast period (2021-2031).
Get Free Sample Copy of this Report@ https://www.persistencemarketresearch.com/samples/4456
In terms of application, the growth factors market can be segmented into tissue engineering, drug development, cell-based therapies, and cosmetics. The tissue engineering segment is expected to hold a significant share of the market during the forecast period, owing to the increasing demand for growth factors in tissue engineering applications. The drug development segment is also expected to witness significant growth during the forecast period, owing to the increasing demand for growth factors in drug development. The cell-based therapies segment is expected to witness moderate growth during the forecast period, owing to the increasing adoption of cell-based therapies and the growing research and development activities related to growth factors. The cosmetics segment is expected to witness significant growth during the forecast period, owing to the increasing demand for growth factors in the cosmetics industry.
According to the World Health Organization (WHO), cancer caused around 9.9 million deaths worldwide in 2020. In addition, around 19.2 million people across the world were suffering from cancer. Also, increasing occurrence of autoimmune disorders is anticipated to drive demand for growth factors.
Rising prevalence of autoimmune diseases and cancer and the benefits of cytokine and growth factors therapy in the treatment of these diseases are expected to complement market growth over the coming years.
Growing research in the field of wound healing and skin regeneration is also expected to favor expansion of the global growth factors market. However, high cost associated with GMP grade growth factors is estimated to hamper market growth.
Acquisitions, collaborations, and partnership agreements are rife in this landscape:
PPD, Inc., a leading clinical research organization, was acquired by Thermo Fisher Scientific in April 2021, expanding the company's service offerings to pharma and biotech customers.
Merck and BioMed X Institute extended their collaboration to continue novel oncology and autoimmunity research in 2021.
In 2021, Lonza and ValenzaBio entered into a manufacturing agreement to rapidly advance VB421, an anti-IGF-1R antibody for autoimmune diseases.
Company Profiles:
Thermo Fisher Scientific
Lonza Group AG
Merck KGaA
General Electronics Company
F. Hoffmann-La Roche Ltd
Applied Biological Materials (abm), Inc.
Abcam plc.
Cell Signaling Technology, Inc.
Meridian Bioscience Inc.
Sartorius CellGenix GmbH
Bio-Techne.
Proteintech Group, Inc.
Miltenyi Biotec
Creative Bioarray
Akron Biotech
Sino Biological Inc.
Repligen corporation
LEADGENE BIOMEDICAL, INC.
PeproTech Inc.
Others
Request for Methodology@ https://www.persistencemarketresearch.com/methodology/4456
Key Takeaways from Market Study
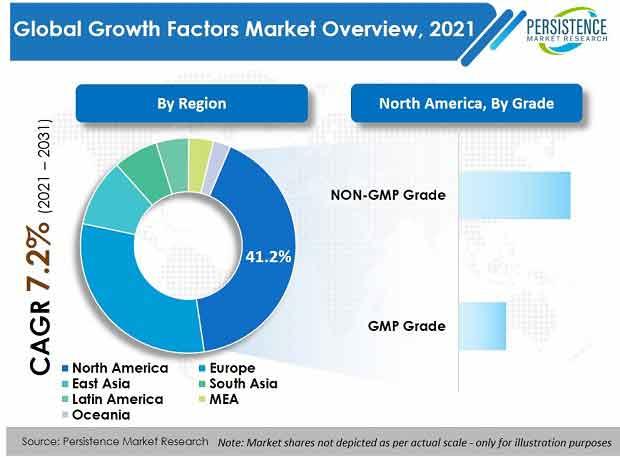
By grade, NON-GMP grade products hold around 70% market value share, globally, primarily due to increasing focus on oncology research and growing contract research activities.
Based on product, interleukins are leading with over 21% market share. Strong product pipelines by various key players are positively driving demand for interleukins among growth factor products.
Cell therapy and ex vivo manufacturing are estimated to dominate the market by application. This segment accounted for approximately 29% share of the market, primarily due to increase in acceptance of growth factors in cancer and autoimmune therapies
Pharmaceutical and biotechnology companies dominate with a market share of 48%. Growing number of R&D activities and increasing preference toward growth factors for gene therapy have had a positive impact on the market.
North America is set to dominate the global market with a value share of around 41%. Europe is slated to be the second-largest region with a value share of 30%.
"Rising prevalence of cancer, preference toward use of regenerative medicines, and new product launches & approvals are expected to drive market growth over the coming years," says an analyst of Persistence Market Research.
Demand-side Drivers:
Increasing focus on oncology research
Growing demand for growth factors and cytokines in stem cell biology
Increasing demand for growth factors in regenerative medicine
Market Opportunities:
Shift toward fast growing Asia Pacific market
Limited number of companies with sole focus on GMP grade products
Market Competition
New product approvals, launches, collaborations, agreements, and partnerships have emerged as the main growth strategy implemented by leading players. By focusing on these strategies, key market players are strengthening their existing product portfolios to expand their geographic footprints.
In 2020, the humankind human cell-expressed growth factor of Proteintech Group, Inc received an ISO 13485 Certificate.
In 2020, Akron Biotech signed an agreement to acquire Cytiva. This acquisition helps enhance its cGMP-compliant solutions and support the development of advanced therapy.
Get up to 20% off on Immediate Purchase @ https://www.persistencemarketresearch.com/checkout/4456
What Does the Report Cover?
Persistence Market Research offers a unique perspective and actionable insights on the growth factors market in its latest study, presenting a historical demand assessment of 2016 - 2020 and projections for 2021 - 2031.
The research study is based on the product (transforming growth factors (TGF), epidermal growth factors (EGFs), platelet-derived growth factors (PDGFs), fibroblast growth factors (FGFs), insulin-like growth factors (IGFs), vascular endothelial growth factors (VEGFs), hepatocyte growth factors (HGFs), tumour necrosis factors (TNFs), interleukins, and others), grade (GMP grade and NON-GMP grade), application (oncology research, haematology research, wound healing research, dermatology research, cardiovascular disease & diabetes, cell therapy and ex vivo manufacturing, and others), and end user (pharmaceutical and biotechnology companies, research centres & academic institutes, and CMOs & CDMOs), across seven key regions of the world.
Know More:
https://www.medgadget.com/2022/05/growth-factors-market-report-projects-revenue-to-surpass-us-3-6-bn-by-2031-endthermo-fisher-scientific-lonza-group-ag-and-more.html
Contact us:
Persistence Market Research
Address - 305 Broadway, 7th Floor, New York City,
NY 10007 United States
U.S. Ph. - +1-646-568-7751
USA-Canada Toll-free - +1 800-961-0353
Sales - sales@persistencemarketresearch.com
About Us:
Persistence Market Research's Expertise in Life Sciences and Transformational Health
Our expert team of industry analysts comprising management graduates, medical professionals, engineers, and project managers provides insights on emerging therapy areas, diagnostic tools, medical devices and components, reimbursement and market access, biotechnology, and life science research products and services to equip decision-makers with sound inputs and strategic recommendations. Click here to learn more about how we zero in on the critical aspects of this industry.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release The Growth Factors Market to look through AI-based genomics at a CAGR of 7.2% here
News-ID: 2853329 • Views: …
More Releases from Persistence Market Research
Active Modified Atmospheric Packaging Market to Surpass US$ 37.9 Bn by 2033, Dri …
The global active modified atmospheric packaging market is entering a dynamic growth phase as food manufacturers, healthcare suppliers, and logistics companies intensify their focus on extending product freshness, minimizing waste, and improving supply chain resilience. Active modified atmospheric packaging (AMAP) integrates advanced gas control technologies, moisture regulators, and antimicrobial features to create optimal internal environments for perishable products.
According to the latest study by Persistence Market Research, the global active modified…

Shunt Capacitor Market Expected to Reach US$2.0 Bn by 2033 Driven by Grid Modern …
The global shunt capacitor market is set for sustained growth as power grids worldwide undergo modernization to meet rising electricity demand and improve energy efficiency. According to the latest study by Persistence Market Research, the global shunt capacitor market size is likely to be valued at US$ 1.3 billion in 2026 and is projected to reach US$ 2.0 billion by 2033, expanding at a CAGR of 6% during the forecast…

Tire Cord & Tire Fabrics Market Set to Hit US$9.0 Bn by 2032 Driven by Radializa …
The global tire cord & tire fabrics market is entering a dynamic growth phase as automotive production rebounds, mobility patterns evolve, and manufacturers prioritize high-performance reinforcement materials. Tire cords and fabrics form the structural backbone of tires, providing dimensional stability, strength, and resistance to wear under demanding operating conditions.
According to the latest study by Persistence Market Research, the market is valued at US$5.9 billion in 2025 and is projected to…

Event Tourism Market Set for Exponential Growth through 2032 - PMR Research
The global Event Tourism Market is poised for remarkable expansion, driven by sustained demand for live experiences, increased business travel, hybrid event adoption, and a rebound in international tourism. According to industry projections, the market is expected to grow from an estimated US$1,538.3 billion in 2025 to US$2,631.5 billion by 2032, registering a CAGR of 7.3% over the forecast period.
This robust growth underscores the evolution of event tourism into one…
More Releases for GMP
Creative Peptides Released GMP Synthesis Service
Located in Shirley, New York, the world’s leading peptide supplier Creative Peptides announced the launch of its GMP synthesis (https://www.creative-peptides.com/services/custom-gmp-peptide-synthesis-services.html ) business on August 29, 2018. Now this company is focused on the development and GMP manufacturing of pharmaceutical grade peptides.
As the demand of pharmaceutical market continues to grow, more and more pharmas and research institutions choose the CMO and CRO models to expand their businesses, which is more…
Diapharm implements European GMP guidelines in China
Münster (DE), London (UK), Ningbo (CN), 20 December 2013 – Pharmaceutical service provider Diapharm (diapharm.com) is increasing its business activities in China: Diapharm has now implemented a “European” quality management system for Neptune Pharma Ltd (www.neptunepharma.com) in their Joint Venture Partner’s factory in Ningbo, Zhejiang Province. And it has done so successfully: The veterinary medicinal product Trident 500mg/g Powder for Suspension for Fish Treatment (www.trident-50.com), is manufactured onsite under EU…
ECA Foundation releases free GMP WebApp
The ECA Foundation has been providing advanced training and information services in the pharmaceutical industry and especially with regard to pharmaceutical Quality Assurance and GMP compliance for more than 10 years. Now the organisation took advantage of its extensive experience to develop a further free of charge service – the new GMP WebApp.
This new GMP WebApp runs on all smartphones and tablet PCs (Apple and Android platforms) and allows users…
GMP Friction Products Awarded ISO 9001:2008
Internationally Recognized Certification Measures Consistency in Process, Procedure and Quality Performance in Manufacture of Friction Materials
AKRON, OH (March 23, 2011) -- GMP Friction Products, a world leader manufacturing powdered metal friction products for clutch plates and brake pads, recently received certification for ISO 9001:2008.
“ISO 9001:2008 signifies we have taken the extra measure of documenting the policies and standards to ensure consistent compliance with our manufacturing processes,” said Jerry Lynch,…
GMP MANUAL Volume 2 - Validation Procedures by Maas & Peither AG – GMP Publish …
GMP Publishing is launching its new GMP MANUAL Volume 2 – Validation Procedures.
The compendium on validation procedures was written by Dr. Doris Borchert, Dr. Peter Bosshard, Dr. Ralph Gomez, Dr. Michael Hiob, Dr. Christine Oechslein, Max Lazar, Ulrike Reuter, Michael Schulte, Uwe Schwarzat – all international experts and key opinion leaders. They share their detailed understanding of the various aspects of the validation process in clear and comprehensive style…
blue inspection body celebrates 50 GMP audits
Münster (Germany), 20 November 2009. Two years after founding the company and just 18 months after gaining the accreditation blue inspection body GmbH announced today the successful execution of its 50th GMP audit. Further audit trips to China, India, Israel and various European countries have been scheduled already, meaning that in the first quarter 2010 the 75th audit is targeted to be completed. Blue, as a privately organised inspection body,…