Press release
Pediatric Clinical Trials Market Overview, Trends, Growth, Demand, Key Players Analysis and Forecast to 2032
According to Future Market Insights, the global pediatric clinical trials Market Share is projected to grow steadily at a 5.3% value CAGR, during the forecast period 2022-2032. Earlier in 2021, this market was projected to have a global market valuation worth US$ 15 Billion and is expected to reach a sum of US$ 26.47 Billion by 2032.Due to unmet healthcare needs in the pediatric patient population, there is a considerable product pipeline in the clinical phase of development. In the foreseeable future, the market will benefit from a robust medication development pipeline. Furthermore, in 2021, there were over 1,300 industry-sponsored pediatric clinical studies underway spanning a wide range of therapeutic categories, including communicable diseases, genetic disorders, neurologic issues, and numerous types of cancer.
Increasing pediatric medicine awareness, outsourcing in-house clinical trials to CROs, and the Increasing Burden of Pediatric Diseases such as Diabetes are projected to drive market growth globally. By region, North America is observed to have a substantial proportion of the pediatric clinical trial market, and this trend is projected to continue during the projection period.
The United States has the region's most lucrative market. The rising prevalence of disorders in the country's pediatric population is strengthening research initiatives to produce particular medications and expanding the number of clinical trials. This, together with other factors, is driving demand for the worldwide pediatric clinical trials market.
Request Sample Report @ https://www.futuremarketinsights.com/reports/sample/rep-gb-15017
Prominent Growth Drivers Influencing Pediatric Clinical Trials Industry
Increased Incidence of Chronic Diseases to accelerate Market Growth
The growing demand for innovative vaccines and biological products to treat infectious illnesses such as measles, polio, mumps, TB, rubella, influenza, whooping cough, chickenpox, malaria, and staphylococcus pharyngitis is likely to fuel market expansion. According to the World Health Organization (WHO) 2021, over 85 percent of infants worldwide are vaccinated against 27 illnesses; yet, 19.4 million children are unvaccinated, resulting in the deaths of 2-3 million children per year. According to the Centers for Disease Control and Prevention (CDC) fact page, in 2021, over 471,000 children in the United States had active epilepsy, accounting for approximately 0.6 percent of children aged 0-17 years.
At the same time, diabetes is a major concern among children nowadays. According to the International Diabetes Federation's (IDF) Diabetes Atlas 2021, around 464 million children globally had diabetes in 2021. One million of these people (children and adolescents under the age of 20) have type 1 diabetes.
Rising parental awareness of pediatric clinical trials for dangerous infections and chronic disorders, as well as a substantial number of CROs performing pediatric studies, are driving the market. Autism is one of the most frequent childhood disorders. According to the CDC, one in every 44 children in the United States will have autism by 2021. The increased prevalence of autism among children is expected to propel the market for autism clinical trials, promoting pediatric clinical market growth globally.
Shifting In-house Clinical Trials to CROs to augment the market growth
The pharmaceutical business has increasingly shifted from in-house clinical research to contract research organizations during the previous decade (CRO). According to the Tufts University in association with ICON PLC on CRO strategic alliances, it is anticipated that by 2022, 72% of all clinical trials would have been moved to CROs worldwide. CROs' roles are becoming more crucial as they get more active in clinical research and are viewed as more considered partners, offering access not only to devoted experts but also to patients all over the world.
Request for Customization @ https://www.futuremarketinsights.com/customization-available/rep-gb-15017
Both the Food and drug administration and the European Medicines Agency (EMA) have made significant revisions to their standards, and most clinical studies now require a pediatric component. The expansion of these pediatric studies has also resulted in the establishment of pediatric CROs, such as Paidion Research, the first global CRO dedicated solely to pediatric health research. Many CROs are merging in order to raise their profiles and improve their services. Synteract, for example, bought Pediatric CRO KinderPharm to bolster its pediatric development center, which provides pharmacometric modeling and clinical trial simulation technology, as well as juvenile formulation and toxicological services. Such advancements in CROs have fueled the practice of outsourcing in-house clinical trials to CROs.
What could possibly hinder Pediatric Clinical Trials Deployment?
Strict Regulations during the Pandemic and Risk among Children will restrict the Market's Growth
Pediatric clinical studies were suspended during the pandemic to limit virus spread. This had an impact on the recruitment of young patients for new clinical studies. Such occurrences are likely to have a detrimental impact on market growth.
The high termination rate of pediatric clinical trials for a variety of reasons is projected to stifle expansion in the pediatric clinical trials market. Pediatric studies have a high failure rate due to factors such as patient accrual. Other concerns include behavioral issues and trial conduct issues such as improper dosing, formulation difficulty, and informative termination, as well as regulatory issues and medication toxicity during clinical studies. According to the US Food and Drug Administration, around 30 to 45% of pediatric trials fail to show safety, efficacy, or result in a labeled indication for pediatric use, resulting in their cancellation.
Differences in the acceptable degree of risk to which children may be exposed in clinical studies are also a problem. The requirement to preserve equipoise within study arms by balancing risks across groups complicates study design, enrollment, and patient consent. When this is done, the choice to enroll a child in a clinical study can be made on the same risk/benefit basis as the non research alternative for care. In reality, regulatory rules in the United States limit the permitted risk exposure for children when there is no recognized direct benefit to them from participating in the study. Furthermore, when direct benefits are possible for a certain study, constraints are placed on the basis for risk exposure. These are going to be some of the serious concerns for the key players in the market.
Phase-II Clinical Trials likely to gain maximum share
Phase II trials category dominates the market, accounting for more than 33% of global revenue. Phase II clinical studies are designed to determine the pharmacological efficacy of new medicines. They are usually single-arm experiments, although they can also be multi-arm trials. Multiple-arm trials with or without control arms might be randomized or nonrandomized.
Children are subjected to phase II trials to examine the intervention's safety and efficacy. Phase II research includes a large number of pediatric individuals; also, studies in this stage are more sophisticated than those in previous phases. Generally, testing of pharmaceutical products in children is postponed until the trials reach phase III, which investigates efficacy, acceptability, and safety.
Speak to our Research Expert @ https://www.futuremarketinsights.com/ask-question/rep-gb-15017
Although the deferral is intended to protect children from harm, it also indicates that children's access to potentially beneficial treatments will be delayed. Phase II trials have been shown to have higher success rates and are increasingly being conducted in the market due to their superior efficacy.
By Study Design, which Category is likely to benefit the most?
The Treatment Studies Segment is forecast to grow in the future
Treatment studies account for more than 66% of the global market's revenue. According to Clinicaltrials.gov, 63,060 pediatric trials were filed between 2001 and 2021, with 37,136 (68.1 percent) containing therapy studies. There were fewer drug studies conducted throughout time, although the majority of therapy pediatric trials were single site (61.4 percent), small-scale (58.9 percent), randomized (66.0 percent), and had no masking (56.9 percent).
Institutions (78.8 percent), industry (19.1 percent), NIH (National Institute of Health) (1.6 percent), and U.S. public agencies fund the majority of pediatric trial treatment studies (0.5 percent). This is one of the primary causes for the segment's rapid growth in the global market.
Which Therapeutic Area will gain prominence during the Forecast Period?
Oncology is expected to be the fastest growing during the forecast period
By therapeutics, oncology is expected to hold a high revenue share of more than 17% of the global market. Cancer is a leading cause of death worldwide. Cancer cases among youngsters under the age of 19 years are expected to reach 291,000 by 2040.
As per the American Cancer Society, an estimated 10,500 children under the age of 15 in the United States will be diagnosed with cancer in 2022. It also claims that pediatric cancer rates have risen in recent decades. Furthermore, According to clinicaltrials.gov, there were roughly 3,287 ongoing pediatric clinical trials for cancer in July 2021. The increased number of these trials is projected to boost the market.
The National Cancer Institute established the Molecular Characterization Initiative for Pediatric Tumors in March 2022. The Initiative is provided by the NCI's Childhood Cancer Data Initiative, which was established to encourage data exchange and the collecting of new data among pediatric oncology experts. With the growing number of pediatric cancer cases globally, many hospitals and institutions are focusing on the development of therapeutic medications, resulting in an increase in the number of pediatric clinical trials worldwide.
What is the Outlook of the North American pediatric clinical trials Market?
Increasing chronic diseases among the children in this region will drive this market
North America led the global pediatric clinical trials market, accounting for 45% of global sales. This is owing to the presence of a large number of pharmaceutical companies and contract research organizations (CROs) performing pediatric studies in the region.
The United States is predicted to have a considerable market share and to exhibit a similar trend during the projection period. According to clinicaltrials.gov, there were more than 1,365 ongoing pediatric clinical trials in the United States as of March 2022, representing a significant fraction of the global number of pediatric research operations. The high number of trials, as well as the high cost per treatment and patient, have had a significant impact on the market in the United States.
The rising prevalence of disorders in the country's pediatric population is strengthening research initiatives to produce particular medications and increasing the number of clinical trials. According to the National Diabetes Statistics Report 2021, an estimated 185,000 American children and adolescents were seen to be suffering from Type 1 diabetes in 2020, and this figure is expected to rise in the future years. This will necessitate rapid demand in this industry to combat these diseases.
Aside from that, public groups are taking active steps to fund pediatric research. For example, the current NIH funding for pediatric research is USD 4 billion. Such measures will contribute to the expansion of the market in this region.
How is the growth of pediatric clinical trials emerging in the APAC region?
Huge population with varied genetic defects in the APAC region to boost the market
Asia Pacific is expected to grow at the fastest rate over the forecast period, with a 25% market share. Due to the simplicity of regulatory compliance, low study expenses, and rapidly growing pediatric patient population, the Asia Pacific region has become a hotspot for conducting clinical trials.
APAC has a genetically varied population of approximately 4 billion individuals, many of whom may be treatment-naive in nations such as India and China. As a result, recruiting pediatric patients who match qualifying criteria may be simpler than in Western countries. Furthermore, APAC countries frequently have a big number of desirable patients who show lifestyle-related health and genetic disorders that were previously more widespread in the Western world. In India, for example, children are still born with a variety of genetic abnormalities and chronic diseases.
Furthermore, due to the high frequency of pediatric disorders, supportive activities boosting pediatric research in the region are propelling the regional market. For example, UNESCO reported in March 2022 that China is focusing on creating improvements in neuroscience to treat children with autism in the country.
Competitive Landscape
Due to the sheer presence of a few important players in the market with very few additional participants, the industry studied is consolidated. Market participants' primary strategies included trial initialization, acquisitions, and collaborations, among others.
Pfizer began a Phase 2/3 research for PAXLOVID tablets, which are especially intended to treat COVID-19 in young patients, in March 2022. PAXLOVID reduced the risk of hospitalization or death from any cause by 89 percent (within three days of symptom start) and 88 percent (within five days of appearance of symptoms).
Novartis announced intentions to begin SMART, a Phase 3b clinical research to assess the safety and efficacy of Zolgensma (onasemnogene abeparvovec) in young children with spinal muscular atrophy, in April 2021. SMART is a multicenter, Phase 3b, single-arm, open-label study designed to assess the safety, tolerability, and efficacy of a one-time IV infusion of Zolgensma in pediatric patients with symptomatic SMA who have bi-allelic genetic changes in the SMN1 gene and any copy number of the SMN2 gene and weigh between 8.5 kg and 21 kg. The study will enroll 24 patients and will monitor them for a 12-month period.
Key Segments Covered in the Pediatric Clinical Trials Market Report
Pediatric Clinical Trials Market by Phase Type:
Phase I Pediatric Clinical Trials
Phase II Pediatric Clinical Trials
Phase III Pediatric Clinical Trials
Phase IV Pediatric Clinical Trials
Pediatric Clinical Trials Market by Study Design:
Treatment Studies of Pediatric Clinical Trials
Observational Studies of Pediatric Clinical Trials
Pediatric Clinical Trials Market by Therapeutic Areas:
Pediatric Clinical Trials for Infectious Diseases
Pediatric Clinical Trials for Oncology
Pediatric Clinical Trials for Autoimmune/Inflammatory Diseases
Pediatric Clinical Trials for Respiratory Disorders
Pediatric Clinical Trials for Mental Health Disorders
Pediatric Clinical Trials for Other Therapeutic Areas
Contact:
Future Market Insights, Inc.
Christiana Corporate, 200 Continental Drive,
Suite 401, Newark, Delaware - 19713, USA
T: +1-845-579-5705
Browse All Reports: https://www.futuremarketinsights.com/reports
About Future Market Insights (FMI)
Future Market Insights (ESOMAR certified market research organization and a member of Greater New York Chamber of Commerce) provides in-depth insights into governing factors elevating the demand in the market. It discloses opportunities that will favor the market growth in various segments on the basis of Source, Application, Sales Channel and End Use over the next 10-years.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Pediatric Clinical Trials Market Overview, Trends, Growth, Demand, Key Players Analysis and Forecast to 2032 here
News-ID: 2751212 • Views: …
More Releases from Future Market Insights
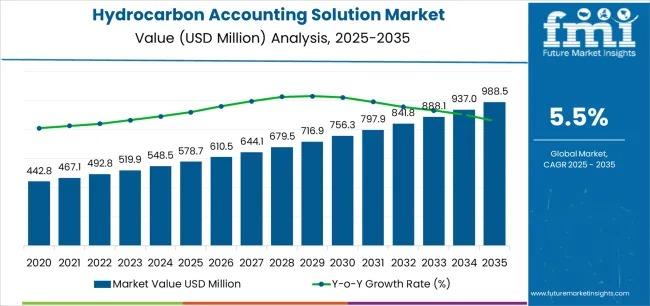
Hydrocarbon Accounting Solution Market Set to Reach USD 988.4 Million by 2035 as …
The global hydrocarbon accounting solution market is projected to expand from USD 578.7 million in 2025 to USD 988.4 million by 2035, registering a steady compound annual growth rate (CAGR) of 5.5%. This expansion reflects a structural shift across the oil and gas industry toward digital platforms capable of delivering accurate production allocation, automated reconciliation, and regulatory compliance in increasingly complex operational environments. As production portfolios expand across unconventional resources,…
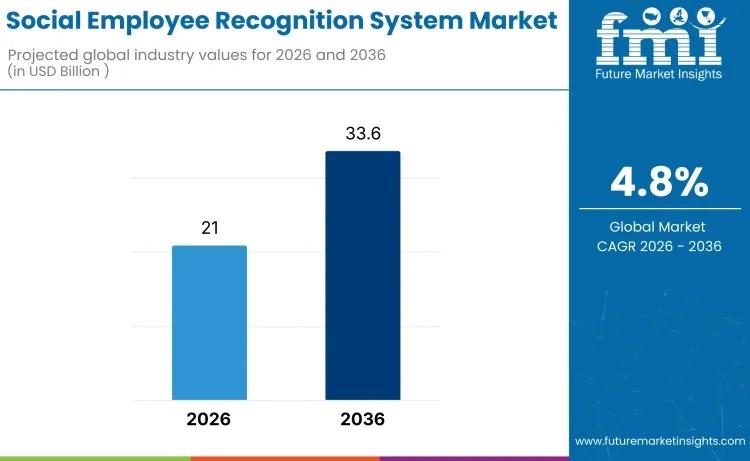
Social Employee Recognition System Market to Reach USD 33.6 Billion by 2036 as D …
The global social employee recognition system market is entering a new phase of strategic expansion, projected to grow from USD 21.0 billion in 2026 to USD 33.6 billion by 2036, reflecting a compound annual growth rate (CAGR) of 4.8%. This sustained growth trajectory highlights the increasing importance of employee engagement technologies in shaping organizational culture, enhancing workforce productivity, and supporting talent retention initiatives across diverse industries.
Organizations worldwide are prioritizing digital…
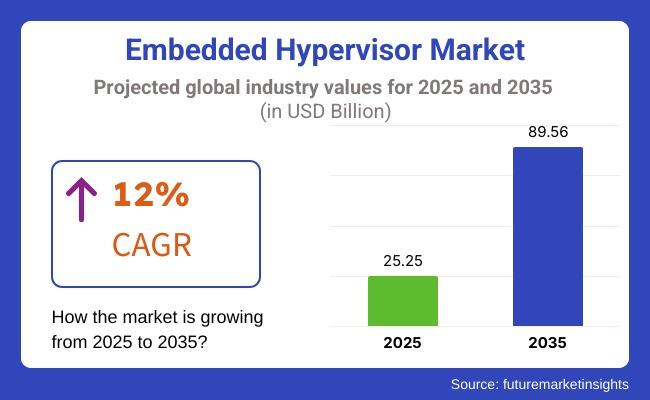
Embedded Hypervisor Market Set for Strong Expansion as Software-Defined Vehicles …
The global embedded hypervisor market is entering a period of accelerated expansion, driven by rising adoption of software-defined architectures across automotive, aerospace, industrial automation, and telecommunications industries. The market is projected to grow from USD 25.25 billion in 2025 to USD 89.56 billion by 2035, advancing at a compound annual growth rate (CAGR) of 12%, reflecting the increasing importance of secure, high-performance virtualization in mission-critical embedded systems.
Embedded hypervisors-specialized software layers…
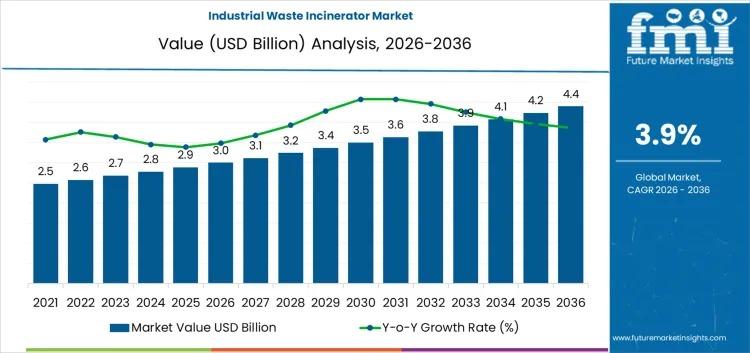
Industrial Waste Incinerator Market to Reach USD 4.4 Billion by 2036 as Industri …
The global industrial waste incinerator market is projected to grow from USD 3 billion in 2026 to USD 4.4 billion by 2036, registering a compound annual growth rate (CAGR) of 3.90% over the forecast period. This steady expansion reflects increasing global pressure on industrial operators to adopt sustainable waste management practices, comply with stringent environmental regulations, and reduce landfill dependence while improving operational efficiency.
As industrial sectors expand production capacity and…
More Releases for Pediatric
Chronic Pediatric Conditions Fuels Growth In The Pediatric Radiology Market: An …
The Pediatric Radiology Market Report by The Business Research Company delivers a detailed market assessment, covering size projections from 2025 to 2034. This report explores crucial market trends, major drivers and market segmentation by [key segment categories].
What Is the Projected Growth of the Pediatric Radiology Market?
The pediatric radiology market, valued at $5.94 billion in 2024, is projected to increase to $6.29 billion in 2025, reflecting a CAGR of 5.9%. Growth…
Pediatric Perfusion Market Experiences Growth with Increasing Pediatric Surgerie …
Market Overview
Pediatric Perfusion Market size was valued at US$ 575.3 Mn in 2025 and is expected to reach US$ 842.5 Mn by 2032 growing at a compound annual growth rate (CAGR) of 5.6% from 2025 to 2032.
Coherent Market Insights has released a comprehensive new report offering an in-depth analysis of the Pediatric Perfusion Market from 2025 to 2032. This study provides reliable global and regional projections, helping stakeholders navigate the…
Adorable Smiles Pediatric Dentistry: Pediatric Dentist Corinth, TX
Adorable Smiles Pediatric Dentistry [https://adorablesmilestx.com/] is proud to provide exceptional dental care for children in Corinth, TX. Conveniently located at 3901 FM 2181 Suite 140, the practice is a trusted choice for families looking for pediatric dental services in Corinth TX. With a fun, child-friendly atmosphere, the team ensures that every visit is enjoyable and stress-free for kids.
"Our goal is to create a positive dental experience for every child, building…
Pediatric Interventional Cardiology Market: Advancements and Opportunities for P …
The report is generically segmented into six parts and every part aims on the overview of the Pediatric Interventional Cardiology Market industry, present condition of the market, feasibleness of the investment along with several strategies and policies. Apart from the definition and classification, the report also discusses the analysis of import and export and describes a comparison of the market that is focused on the trends and development.
Along with…
Pediatric Neuroblastoma Treatment Market - Redefining Possibilities: Advancement …
Newark, New Castle, USA: The "Pediatric Neuroblastoma Treatment Market" provides a value chain analysis of revenue for the anticipated period from 2023 to 2031. The report will include a full and comprehensive analysis of the business operations of all market leaders in this industry, as well as their in-depth market research, historical market development, and information about their market competitors
Pediatric Neuroblastoma Treatment Market: https://www.growthplusreports.com/report/pediatric-neuroblastoma-treatment-market/7681
This latest report researches the industry structure,…
Global Pediatric Vaccines Market | Global Pediatric Vaccines Industry | Global P …
The pediatric vaccines market involves of sales of pediatric vaccines and its linked services utilization to provide immunity to infants and children for specific syndromes. Pediatric vaccine is a preparation of killed the microorganisms, living tempered organisms, or living fully virulent organisms that are controlled to encourage the production of antibodies and deliver the immunity against one or numerous diseases among children. Pediatric vaccines are utilized in childhood immunization schedules…