Press release
OptraSCAN and Inspirata Announce Their Technical Partnership to Strengthen Digital Pathology Adoption
SAN JOSE, Calif., Nov. 9, 2021 /PRNewswire/ --OptraSCAN®, a leading end-to-end digital pathology solution provider announces its partnership with Inspirata Inc., a leading provider of digital pathology and cancer informatics solutions. This partnership will allow pathology labs, healthcare networks, and hospitals to acquire digital pathology images without any upfront cost and display them within a world-class workflow solution."We are excited to be working with Inspirata to support their mission to digitize and automate the entire oncologic pathology workflow," said Abhi Gholap, Founder of OptraSCAN. "Our On-Demand scanning model will allow pathologists to digitize their slides without any capital cost," he further added.
OptraSCAN's partnership with Inspirata marks the latest extension to its affordable pay-per-use scanning devices. Shared Inspirata and OptraSCAN customers can now utilize native OptraSCAN image files from within Dynamyx™. Facilitating slide digitization at an affordable cost, the seamless integration of OptraSCAN provides:
• Cloud-based whole slide imaging systems.
• Flexibility to choose from 15 slides capacity to 480 slides capacity.
• Well-equipped for confocal, brightfield, fluorescence, and frozen section applications.
"Inspirata is delighted to work with its friends at OptraSCAN to accelerate global adoption of digital pathology. OptraSCAN's status as an approved partner guarantees the seamless integration of its leading whole slide image scanners with Inspirata's Dynamyx™ digital pathology workflow software," said Dr Mark Lloyd, Founder and EVP, Inspirata.
“Following our recent successes with Inspirata’s Dynamyx™ digital pathology workflow software in Europe, Fujifilm welcomes this partnership between Inspirata and OptraSCAN,” said Tim Wing, Head of Digital Pathology at Fujifilm Europe. “The OptraSCAN range fits small low volume specialist labs all the way up to high volume multi-site labs and will further increase the options for customers of the Fujifilm Digital Pathology Solution looking to make informed choices regarding scanners that fit their often-unique requirements. OptraSCAN’s digital pathology scanners paired with Inspirata’s open-architecture Dynamyx™ case management platform will allow pathologists to implement digital pathology solutions at economical pricing and enable healthcare providers to arrive at their preferred blend of laboratory and diagnostic technologies.”
For more information visit: https://www.optrascan.com/
OptraSCAN® whole slide scanners are CE marked for IVD use.
OptraSCAN® is an ISO13485 certified company.
About Inspirata, Inc.
Inspirata helps patients fighting cancer—and the clinicians they trust—to make every moment matter. Our comprehensive cancer informatics solutions bring disparate data together throughout the entire cancer care journey to drive informed decisions that improve survivorship.
Inspirata has assembled the most advanced and proven technologies to address the complex challenges of delivering cancer care and conducting ground-breaking research. We combine leading digital pathology solutions with automated cancer registry solutions, comprehensive cancer informatics and advanced patient engagement tools to bring users the broadest oncology informatics platform available globally. To learn more visit www.inspirata.com.
Dynamyx is CE marked for IVD use.
Dynamyx is USFDA 510(k) pending.
OptraSCAN Contacts:
Anjana Athanikar
Senior Product Manager
Email: a.athanikar@optraventures.com
Tel: +91.20.66540900 x 231
Inspirata Contact:
Emil Mladenov
Vice President of Corporate and Digital Marketing
E-mail: emladenov@inspirata.com
Tel: +1 813-467-7616
1798 Technology Drive, Suite 244, San Jose, CA 95110, USA.
OptraSCAN® are pioneers in the On-Demand Digital Pathology® System, focused on delivering fully integrated, affordable solutions that will maximize your return on investment and improve the performance of your pathology services. OptraSCAN is working to eliminate the barriers to "Go Digital" no matter the size of the pathology lab, the lab's throughput, or global location.
OptraSCAN's end-to-end digital pathology solution provides effective acquisition of whole slide images, viewing, storing, real-time sharing, reporting, and AI & ML based Image analysis solutions via On-Demand or outright purchase model. Follow Us on LinkedIn and Twitter.
OptraSCAN Systems are for research use only in North America.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release OptraSCAN and Inspirata Announce Their Technical Partnership to Strengthen Digital Pathology Adoption here
News-ID: 2456660 • Views: …
More Releases from OptraSCAN Inc
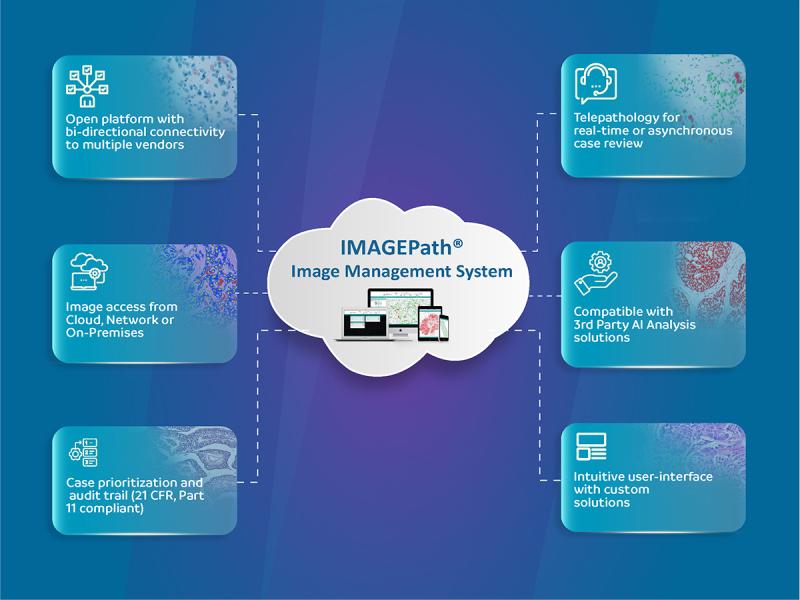
OptraSCAN Launches a Comprehensive Image Management and Image Storage Solution w …
SAN JOSE, Calif., March 12, 2024 /PRNewswire/ -- OptraSCAN®, the leading end-to-end digital pathology solution provider, announces the introduction of a combined Image Management and Image Storage Solution, to enable and simplify the digital transformation of pathology laboratories. The OptraSCAN IMAGEPath™ image management system is an open platform that can easily integrate with scanners from multiple manufacturers and over 20 different image formats, in addition to being compatible with external…

OptraSCAN Empowers AIG Hospitals with Digital Pathology Solutions
Hyderabad, July 4th, 2023 - OptraSCAN, a global leader in digital pathology solutions, is delighted to announce the successful implementation and utilization of its high-speed Digital Pathology Platform OS-Ultra by AIG Hospitals (A unit of Asian Institute of Gastroenterology), Hyderabad. This partnership marks another milestone in OptraSCAN's commitment to revolutionizing the field of digital pathology and empowering healthcare organizations worldwide.
AIG Hospitals, a renowned multidisciplinary institution headed by Chairman Dr. Duvvur…

OptraSCAN Launches OnDemand Digital Pathology
Don't Buy, Just Subscribe
OptraSCAN®, the leading end-to-end digital pathology solution provider announces the introduction of OnDemand Digital Pathology® solution to meet today's need for complete digital transformation of pathology laboratories in North America. Laboratories and Hospitals can save hundreds of thousands of dollars spent on Capital purchases by subscribing to OptraSCAN OnDemand Digital Pathology® for a low monthly fee.
OptraSCAN OnDemand Digital Pathology® subscription service will offer OptraSCAN whole slide digital…

Paige and OptraSCAN Partner to Streamline Digital Pathology Adoption
NEW YORK--Paige, a global leader in end-to-end digital pathology solutions and clinical AI applications, and OptraSCAN®, a leading producer of whole slide scanners, today announced a partnership to streamline the adoption of digital pathology technology in the United States, European Union countries and United Kingdom. The companies aim to reduce barriers to digitizing glass pathology slides, which will in turn enhance workflows in pathology and oncology.
Together, the companies will integrate…
More Releases for Inspirata
Digital Pathology Market Detailed In New Research Report 2024 | Sectra AB, Propa …
The latest market intelligence report published by CMI with the title "Global Digital Pathology Market 2024, Growth Opportunities, and Forecast" provides actionable insights on Medical Devices industry. The report provides demand analysis, industry insights, competitive intelligence, and customer database.
The Research report on Digital Pathology Market presents a complete judgment of the market through strategic insights on future trends, growth factors, supplier landscape, demand landscape, Y-o-Y growth rate, CAGR, pricing analysis.…
Whole Slide Imaging Market Worth $1678.6 million by 2030: Players 3DHISTECH Ltd. …
As per the report published by The Brainy Insights, The global whole slide imaging market is expected to grow from USD 408.6 million in 2021 to USD 1678.6 million by 2030, at a CAGR of 17% during the forecast period 2022-2030. Asia Pacific region emerged as the largest market for the global whole slide imaging market, with a 38% share of the market revenue in 2021. The Asia Pacific region…
Oncology Informatics Market 2023-2030 Growth, Analysis Report, Share, Trends | T …
Infinity Business Insights has recently released a comprehensive research report titled "Oncology Informatics Market Insights, Extending to 2030." This publication spans over 110+ pages and offers an engaging presentation with visually appealing tables and charts that are self-explanatory. The worldwide Oncology Informatics market is expected to grow at a booming CAGR of 12.2% during 2023-2030. It also shows the importance of the Oncology Informatics market main players in the sector, including…
Digital Pathology Market 2023 | Newest Industry Data, Future Trends And Forecast …
Newark, New Castle, USA - new report, titled Digital Pathology Market The report has been put together using primary and secondary research methodologies, which offer an accurate and precise understanding of the Digital Pathology market. Analysts have used a top-down and bottom-up approach to evaluate the segments and provide a fair assessment of their impact on the global Digital Pathology market. The report offers an overview of the market, which…
Whole Slide Imaging Market 2022 Future Scope and SWOT Analysis by 2030 - 3DHISTE …
The study based on the Global Whole Slide Imaging Market published by The Brainy Insights provides an up-to-date and accurate market picture. It also provides important data on the various components which affect the progress of the Whole Slide Imaging market through a brief scan and detailed information. The report will help stakeholders and partners define the best development methods and take advantage of the opportunities prevailing in the Whole Slide Imaging…
Clinical Data Analytics in Healthcare Market May Set New Growth Story | Inspirat …
A Latest intelligence report published by AMA Research with title "Clinical Data Analytics in Healthcare Market Outlook to 2026. A detailed study accumulated to offer Latest insights about acute features of the Global Clinical Data Analytics in Healthcare Market. This report provides a detailed overview of key factors in the Clinical Data Analytics in Healthcare Market and factors such as driver, restraint, past and current trends, regulatory scenarios and technology…