Press release
OptraSCAN Announces CytoSiA – A Complete Digital Solution For Scanning And Analysis Of Cytology Slides At Affordable Pricing
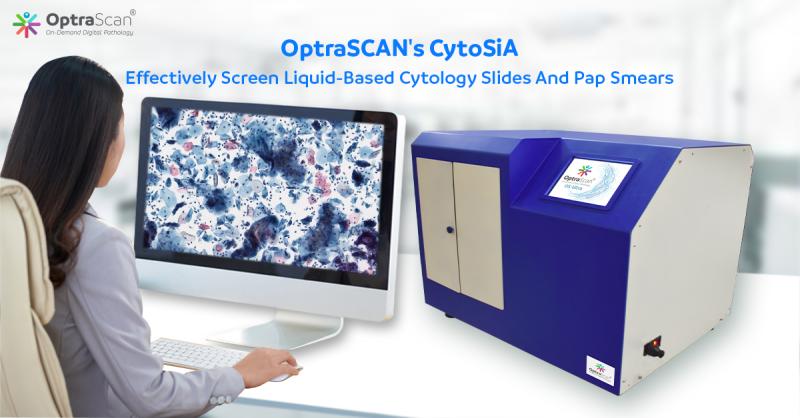
San Jose, California – OptraSCAN®, the leading On-Demand Digital Pathology solution provider, today announced its intelligent solution CytoSiA for rapid yet affordable scanning and analyzing of liquid-based cytology slides and pap-smears. It is a complete solution consisting of OptraSCAN’s digital pathology scanner, storage, and powerful artificial intelligence (AI) algorithms to assist pathologists and cytotechnologists in screening and detection of cervical cancer, pre-cancerous lesions, atypical cells, and all other cytologic categories. Multiple hospitals and pathology laboratories globally have already installed CytoSiA and are witnessing improved patient outcomes, increased efficiency, and productivity needed to cope with the ever-increasing demand of cytology cases.“OptraSCAN is redefining cervical cancer screening by introducing CytoSiA, it offers a plethora of features, to effectively screen liquid-based cytology slides, and pap smears to differentiate between normal and abnormal cervical cells using Bethesda classifications on normal to squamous cell carcinoma (NILM/LSIL/HSIL/SCC)”, said Abhi Gholap, Founder of OptraSCAN. With a range of scanning devices handling as low as 50 slides a week to 5000 slides a week at an extremely affordable price,” he further added.
OptraSCANS’s digital scanners can scan cytology slides of size 15x15 mm area at 40x magnification in less than 60 seconds while generating the highest quality image. CytoSia incorporates patented technology- composite imaging which finds all pixels in focus from various Z plane images and stitches back to create a single layer composite image. This composite imaging technology is considerably efficient as compared to traditional Z-Stacking Technology. It allows for rapid and precise screening of the entire slide within seconds. This level of precision and quality is imperative for analyzing the samples. Furthermore, it presents to the screener images of fields that would be essential in providing a cytological interpretation, place them into categories, and filter out the redundancies.
CytoSiA when used as a companion diagnostic tool it notifies pathologists when inconsistencies between their interpretation and the AI algorithm's findings are observed, offering a safeguard against error or misinterpretation, while also improving overall care quality.
For more information visit: https://www.optrascan.com/solutions/cytosia
Features of CytoSiA:
• Automated computation of sample adequacy for the whole slide cytology image
• Identification of abnormal cells and other entities based on morphological features and AI-based classification using Bethesda scoring
• Identification of reactive, endometrial, actinomyces, candida, clue cells, trichomonas vaginalis, and herpes entities
• Identification of entities including blood, inflammation, and lubricant.
“AI-based image analysis requires images that are of superior quality,” said Dr. Aparna Joshi, Medical Director, at OptraSCAN. “Our team has developed an intelligent imaging technology that transforms physical glass cytology slides into digital images with exceptional clarity. Advanced image analysis and standardization are now achievable with this quality of digitization”, she further added.
Visit- https://www.optrascan.com/
1798 Technology Drive, Suite 244, San Jose, CA 95110
OptraSCAN® are pioneers in the On-Demand Digital Pathology® System, focused on delivering fully integrated, affordable solutions that will maximize your return on investment and improve the performance of your pathology services. An ISO 13485 certified company and CE-marked whole slide scanners for IVD use, OptraSCAN are working to eliminate the barriers to “Go Digital” no matter the size of the pathology lab, the lab’s throughput, or global location.
OptraSCAN’s end-to-end digital pathology solution provides effective acquisition of whole slide images, viewing, storing, real-time sharing, reporting, and AI & ML based Image analysis solutions via On-Demand or outright purchase model. Follow Us on LinkedIn and Twitter.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release OptraSCAN Announces CytoSiA – A Complete Digital Solution For Scanning And Analysis Of Cytology Slides At Affordable Pricing here
News-ID: 2377850 • Views: …
More Releases from OptraSCAN Inc
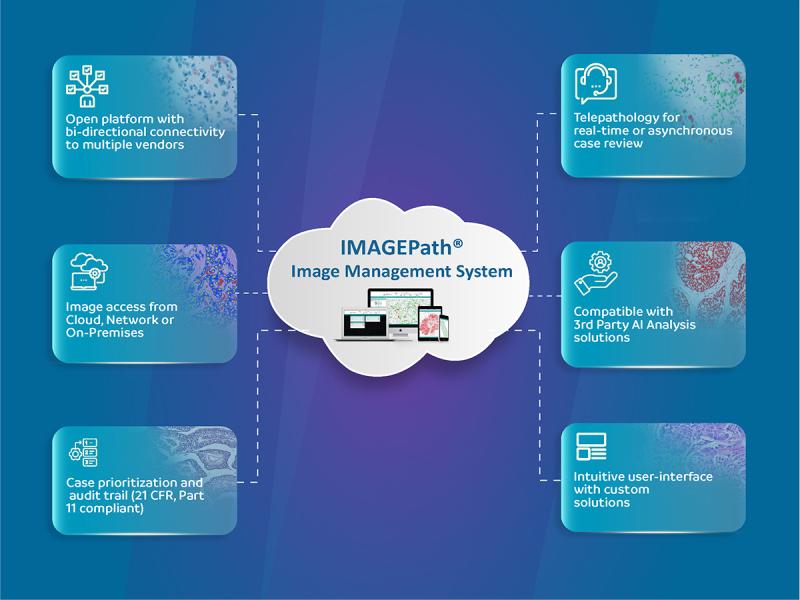
OptraSCAN Launches a Comprehensive Image Management and Image Storage Solution w …
SAN JOSE, Calif., March 12, 2024 /PRNewswire/ -- OptraSCAN®, the leading end-to-end digital pathology solution provider, announces the introduction of a combined Image Management and Image Storage Solution, to enable and simplify the digital transformation of pathology laboratories. The OptraSCAN IMAGEPath™ image management system is an open platform that can easily integrate with scanners from multiple manufacturers and over 20 different image formats, in addition to being compatible with external…

OptraSCAN Empowers AIG Hospitals with Digital Pathology Solutions
Hyderabad, July 4th, 2023 - OptraSCAN, a global leader in digital pathology solutions, is delighted to announce the successful implementation and utilization of its high-speed Digital Pathology Platform OS-Ultra by AIG Hospitals (A unit of Asian Institute of Gastroenterology), Hyderabad. This partnership marks another milestone in OptraSCAN's commitment to revolutionizing the field of digital pathology and empowering healthcare organizations worldwide.
AIG Hospitals, a renowned multidisciplinary institution headed by Chairman Dr. Duvvur…

OptraSCAN Launches OnDemand Digital Pathology
Don't Buy, Just Subscribe
OptraSCAN®, the leading end-to-end digital pathology solution provider announces the introduction of OnDemand Digital Pathology® solution to meet today's need for complete digital transformation of pathology laboratories in North America. Laboratories and Hospitals can save hundreds of thousands of dollars spent on Capital purchases by subscribing to OptraSCAN OnDemand Digital Pathology® for a low monthly fee.
OptraSCAN OnDemand Digital Pathology® subscription service will offer OptraSCAN whole slide digital…

Paige and OptraSCAN Partner to Streamline Digital Pathology Adoption
NEW YORK--Paige, a global leader in end-to-end digital pathology solutions and clinical AI applications, and OptraSCAN®, a leading producer of whole slide scanners, today announced a partnership to streamline the adoption of digital pathology technology in the United States, European Union countries and United Kingdom. The companies aim to reduce barriers to digitizing glass pathology slides, which will in turn enhance workflows in pathology and oncology.
Together, the companies will integrate…
More Releases for CytoSiA
Anatomic Pathology Market Increasing Demand, Growth Analysis and Future Outlook …
The global anatomic pathology market is anticipated to grow at a considerable CAGR of 6.2% during the forecast period. The factor that drives the global anatomic pathology market is product innovation and ongoing R&D activities to develop advanced technologies that assisted in boosting the growth of the market. Various strategies and collaborations are being adopted by these key players to maintain their market share. The key players are found implementing…
Anatomic Pathology Market Trends, Segmentation, Growth, Analysis and Forecast 20 …
The global anatomic pathology market is anticipated to grow at a considerable CAGR of 6.2% during the forecast period. The factor that drives the global anatomic pathology market is product innovation and ongoing R&D activities to develop advanced technologies that assisted in boosting the growth of the market. Various strategies and collaborations are being adopted by these key players to maintain their market share. The key players are found implementing…
Anatomic Pathology Market Trends, Future Outlook, COVID-19 Impact Analysis, Fore …
The global anatomic pathology market is anticipated to grow at a considerable CAGR of 6.2% during the forecast period (2022-2028). The factor that drives the global anatomic pathology market is product innovation and ongoing R&D activities to develop advanced technologies that assisted in boosting the growth of the market. Various strategies and collaborations are being adopted by these key players to maintain their market share. The key players are found…