Press release
OS-Ultra 320 First Ever High-Speed Scanner-OptraSCAN Inc
Unparalleled Image Quality Within SecondsSAN JOSE, Calif- OptraSCAN®, the leading end-to-end digital pathology solution provider, announced the launch of OS-Ultra 320 the world’s first-ever affordable high-speed digital pathology scanner. The OS-Ultra 320 scans tissues/cells of size 15x15 mm area at 40x magnification in less than 60 seconds and features no-touch continuous loading operation. The OS-Ultra 320 is designed to reduce workflow errors and scale up pathology laboratory operations.
“We are disrupting digital transformation of Pathology and Cytology through affordability,” said Abhi Gholap, Founder of OptraSCAN. “With our 15-slide scanner getting tremendous traction for small laboratories, we are happy to launch our high performance 320 slide scanning device at an economical pricing as a complete solution to scan-store-analyze-share the cases,” he further added.
“OS-Ultra 320 represents a significant development in reducing technician time while maintaining perfect image quality for the healthcare professionals. It will provide better performance and improved capabilities,” said Dr. Clive Taylor, Chief Medical Officer of OptraSCAN.
OS-Ultra Technical Specifications:
• Continuous Slide Loading: Intuitive interface with the fully automated walk-away acquisition
• Light Source: LED
• Focusing Technique: Real-Time Auto Focus
• File Format: JPEG2000, TIFF ability to convert file formats
• Slide Formats: Standard 25x75mm (1"x3") slides, 50x75mm (2"x3") slides
• Slide Tolerances: Length: ± 1mm, Width: ± 1mm, Thickness ± 0.2mm
• Resolution: 0.25 μ /pixel
• Workflow Software application
• TELEPath application
• Image Analysis Suite for Breast, Prostate, Brain, Renal, and Lung (IHC and H&E)
For more information visit: https://www.optrascan.com/products/os-ultra-high-speed-whole-slide-scanner
OS-Ultra is also available for purchase via our partners Nikon USA, FujiFilm, and our other distributors - https://www.optrascan.com/contact-us
For scheduling demo, please get in touch at info@optrascan.com
1798 Technology Drive, Suite 244, San Jose, CA 95110
OptraSCAN® are pioneers in the On-Demand Digital Pathology® System, focused on delivering fully integrated, affordable solutions that will maximize your return on investment and improve the performance of your pathology services. An ISO 13485 certified company and CE-marked whole slide scanners for IVD use, OptraSCAN is working to eliminate the barriers to "Go Digital" no matter the size of the pathology lab, the lab’s throughput or global location.
OptraSCAN’s end-to-end digital pathology solution provides effective acquisition of whole slide images, viewing, storing, real-time sharing, reporting and AI & ML based Image analysis solutions via On-Demand or outright purchase model. Follow Us on LinkedIn and Twitter.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release OS-Ultra 320 First Ever High-Speed Scanner-OptraSCAN Inc here
News-ID: 2329314 • Views: …
More Releases from OptraSCAN Inc
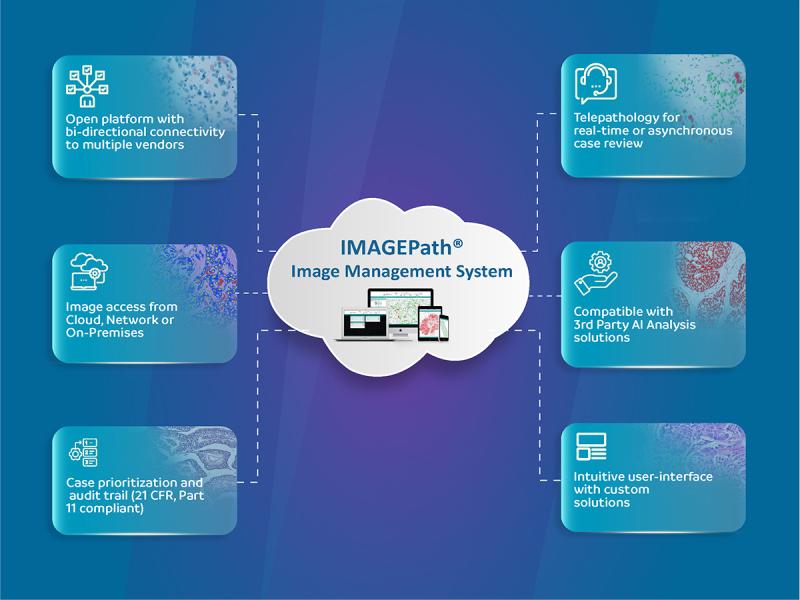
OptraSCAN Launches a Comprehensive Image Management and Image Storage Solution w …
SAN JOSE, Calif., March 12, 2024 /PRNewswire/ -- OptraSCAN®, the leading end-to-end digital pathology solution provider, announces the introduction of a combined Image Management and Image Storage Solution, to enable and simplify the digital transformation of pathology laboratories. The OptraSCAN IMAGEPath™ image management system is an open platform that can easily integrate with scanners from multiple manufacturers and over 20 different image formats, in addition to being compatible with external…

OptraSCAN Empowers AIG Hospitals with Digital Pathology Solutions
Hyderabad, July 4th, 2023 - OptraSCAN, a global leader in digital pathology solutions, is delighted to announce the successful implementation and utilization of its high-speed Digital Pathology Platform OS-Ultra by AIG Hospitals (A unit of Asian Institute of Gastroenterology), Hyderabad. This partnership marks another milestone in OptraSCAN's commitment to revolutionizing the field of digital pathology and empowering healthcare organizations worldwide.
AIG Hospitals, a renowned multidisciplinary institution headed by Chairman Dr. Duvvur…

OptraSCAN Launches OnDemand Digital Pathology
Don't Buy, Just Subscribe
OptraSCAN®, the leading end-to-end digital pathology solution provider announces the introduction of OnDemand Digital Pathology® solution to meet today's need for complete digital transformation of pathology laboratories in North America. Laboratories and Hospitals can save hundreds of thousands of dollars spent on Capital purchases by subscribing to OptraSCAN OnDemand Digital Pathology® for a low monthly fee.
OptraSCAN OnDemand Digital Pathology® subscription service will offer OptraSCAN whole slide digital…

Paige and OptraSCAN Partner to Streamline Digital Pathology Adoption
NEW YORK--Paige, a global leader in end-to-end digital pathology solutions and clinical AI applications, and OptraSCAN®, a leading producer of whole slide scanners, today announced a partnership to streamline the adoption of digital pathology technology in the United States, European Union countries and United Kingdom. The companies aim to reduce barriers to digitizing glass pathology slides, which will in turn enhance workflows in pathology and oncology.
Together, the companies will integrate…
More Releases for Digital
Digital luxury brands Market Is Booming Worldwide | Major Giants Balenciaga Digi …
HTF MI recently introduced Global Digital luxury brands Market study with 143+ pages in-depth overview, describing about the Product / Industry Scope and elaborates market outlook and status (2025-2033). The market Study is segmented by key regions which is accelerating the marketization. At present, the market is developing its presence.
Major companies in Digital luxury brands Market are:
Balenciaga Digital, Louis Vuitton Digital, Gucci Digital, Dolce & Gabbana DGFamily, Prada Virtual,…
Introducing Digital Sales Pro, Inc: Revolutionizing Digital Sales in the Digital …
Digital Sales Pro, Inc. is a company that helps content creators and publishers make money from content and reach a larger audience with their craft.
In today's world, it can be tough for content creators and publishers to make money and connect with their audience.
At, Digital Sales Pro, Inc. we understand these challenges and have created a suite of solutions that help our clients build a strong online presence, monetize…
Digital Twin in Healthcare Market Analysis By Type - Product Digital Twins, Proc …
Introduction
The healthcare industry has seen significant growth and development over the years, with technology playing a critical role in transforming patient care. One such innovative technology that has emerged in recent years is the Global Digital Twin in Healthcare Market. This technology allows healthcare professionals to create a virtual replica of a patient's physical self, enabling them to monitor and analyze patient data in real-time. The Global Digital Twin in…
Digital Twin in Healthcare Market Analysis By Type - Product Digital Twins, Proc …
In 2021, the market for Digital Twin in Healthcare worldwide was worth $6.75 billion US dollars. AMR Group projects that the market will reach US$ 96.5 billion by 2031, growing at a CAGR of 40 percent between 2022 and 2031.
Industry Overview
Digital twins are virtual copies of physical objects or things that data scientist & IT professionals can use to compute simulations prior to developing and deploying the original devices. Digital…
Digital Therapeutics Market, Digital Therapeutics Market Size, Digital Therapeut …
The global digital therapeutics market is expected to reach US$ 8,941.1 Mn by 2025 from US$ 1,993.2 Mn in 2017. The market is estimated to grow with a CAGR of 20.8% during the forecast period from 2018 to 2025.
North America is the largest geographic market and it is expected to be the largest revenue generator during the forecast period, whereas the market is expected to witness growth at a significant…
Digital Display Market Future Growth with Worldwide Players (Digital Virgo, Digi …
Digital Display Industry 2019 Global Market 2025 research report represents the historical overview of current Market situation, size, share, trends, growth, supply, outlook and manufacturers with detailed analysis. It also focuses on Digital Display volume and value at global level, regional level and company level. From a global perspective, this report represents overall Digital Display market size by analyzing historical data and future prospect.
Get Sample Copy of this Report -…
