Press release
Chronic Kidney Disease Drugs Market Analysis Report: Reveals Lookout at Biggest Trends Hitting the Industry Over the Coming Years 2020-2025
The research study covers the current scenario and growth prospects of the Chronic Kidney Disease Drugs market (2020-2025) according to the analysis . This study includes a extensive analysis of the key segments of the industry and also the regional analysis that are taken place throughout the country.Global Chronic Kidney Disease Drugs Market Overview:
A recent report by Business Industry Reports offers an in-depth analysis of global Chronic Kidney Disease Drugs market from 2020 to 2025. It also edifies various sides such as key drivers, prominent developments, and various market opportunities for businesses to gain maximum profit in the projected period.
The presented study elucidates the key indicators of market growth that includes a thorough analysis of the value chain, CAGR growth, and Porter’s Five Forces Analysis. This data will enable readers to understand the quantitative growth parameters of the global Chronic Kidney Disease Drugs market. This Chronic Kidney Disease Drugs market report forecasts revenue growth at global, regional & country levels and provides an analysis on the industry trends in each of the sub-segments from 2020 to 2025. For the purpose of this study, BIR has segmented the global Chronic Kidney Disease Drugs market on the basis of product, application, and region:
The report states that the Chronic Kidney Disease Drugs market has consolidated dynamics and is dominated by a handful of players across the globe. Analysts use the most recent primary and secondary research techniques and tools to arrange comprehensive and accurate marketing research reports. The research study also includes other types of analysis such as qualitative and quantitative. All proportion of shares and breakdowns are determined using secondary sources and verified primary sources. The report could be a collection of first-hand info, qualitative and quantitative assessment by industry analysts, and industry participants across the value chain.
Available Exclusive Sample Copy of this Report @ https://www.businessindustryreports.com/sample-request/231939 .
Market Key Players
The global Chronic Kidney Disease Drugs market is marked by some strong competition from the major players operating in this industry. Many merger and acquisition, joint venture and partnership agreement, product innovation, research and development and geographical extension are some of the key strategies adopted by this player to ensure long term sustenance in these market key participants in the global Chronic Kidney Disease Drugs industry include Keryx Biopharmaceuticals, Inc, Kissei Pharmaceutical Co., Ltd, AbbVie, GlaxoSmithKline, Sanofi, F. Hoffmann-La Roche Ltd, Pfizer…more
Segmentations
The Chronic Kidney Disease Drugs market has been sub-grouped into type and application. The report studies these subsets with respect to the geographical segmentation. The strategists can gain a detailed insight and devise appropriate strategies to target specific market. This detail will lead to a focused approach leading to identification of better opportunities.
Product Type Segmentation : ACE Inhibitors, Calcium Channel Blockers, Beta Blockers, Others
Industry Segmentation : Hospitals, Specialty Clinics
Regional Segmentation
North America (U.S. and Canada)
Latin America (Mexico, Brazil, Peru, Chile, and others)
Western Europe (Germany, U.K., France, Spain, Italy, Nordic countries, Belgium, Netherlands, and Luxembourg)
Eastern Europe (Poland and Russia)
Asia Pacific (China, India, Japan, ASEAN, Australia, and New Zealand)
Middle East and Africa (GCC, Southern Africa, and North Africa)
Purchase this report online with 90 Pages, List of Tables & Figures and in-depth Table of Contents on “Global Chronic Kidney Disease Drugs Report 2020” @ https://www.businessindustryreports.com/buy-now/231939/single .
Global Industry News:
Pfizer : September 23, 2020 FDA ACCEPTS SUPPLEMENTAL NEW DRUG APPLICATION FOR PFIZER’S XALKORI® (CRIZOTINIB) FOR THE TREATMENT OF PEDIATRIC ALK-POSITIVE ANAPLASTIC LARGE CELL LYMPHOMA
Pfizer Inc. today announced that the U.S. Food and Drug Administration (FDA) has accepted and granted priority review to the Company’s supplemental New Drug Application(sNDA)for XALKORI® (crizotinib) for the treatment of pediatric patients with relapsed or refractory systemic anaplastic large cell lymphoma (ALCL) that is anaplastic lymphoma kinase (ALK)-positive. XALKORI received Breakthrough Therapy designation (BTD) for the ALK-positive ALCL indication in May 2018 and if approved, would be the first biomarker-driven therapy for this type of pediatric lymphoma. The Prescription Drug User Fee Act (PDUFA) goal date for a decision by the FDA is January 2021.
“Despite high survival rates for children with ALK-positive anaplastic large cell lymphoma, many will relapse, requiring novel treatment approaches,” said Chris Boshoff, M.D., Ph.D., Chief Development Officer, Oncology, Pfizer Global Product Development. “Today’s FDA filing exemplifies Pfizer’s commitment to broadening the use of biomarker-driven therapies in areas with significant needs, such as rare, pediatric cancers. Given XALKORI’s proven efficacy in ALK-positive lung cancer and activity seen in clinical trials investigating relapsed or refractory ALK- and ROS-1 positive anaplastic large cell lymphoma, if approved, XALKORI could represent an important step toward improving outcomes for children with this type of cancer.”
Anaplastic large cell lymphoma is a rare type of non-Hodgkin lymphoma (NHL), divided into ALK-positive or ALK-negative disease. Though the five-year survival rate for children with cancer in the U.S. is now the highest it’s ever been at 80%, children with cancer continue to face challenges in treating their disease, including rare tumor types, variations in medicine response and prolonged risk of side effects.
“Children with cancer and their families have been desperate for new and innovative medicines. We applaud Pfizer’s commitment to drug discovery and development to help address childhood cancers, and continue to work together on behalf of patients with these cancers,” said George Dahlman, Chief Executive Officer, Children’s Cancer Cause. “We look forward to potentially having a new biomarker-driven therapy for children with relapsed or refractory ALK-positive ALCL that may help improve these children’s lives.”
Key Points Covered :
This research study inspects the current market trends related to the demand, supply, and sales, in addition to the recent developments. Major drivers, restraints, and opportunities have been covered to provide an exhaustive picture of the market. The analysis presents in-complexity information regarding the development, trends, and industry policies and guidelines implemented in each of the geographical regions. Further, the overall directing framework of the market has been thoroughly covered to offer stakeholders a better understanding of the key factors affecting the overall market environment.
The study is a source of reliable data on:
Market segments and sub-segments
Market trends and dynamics
Supply and demand
Market size
Current trends/opportunities/challenges
Competitive landscape
Technological breakthroughs
Value chain and stakeholder analysis
Grab Your Report at an Impressive Discount @ https://www.businessindustryreports.com/check-discount/231939 .
Major Points in Table of Contents:
Global Chronic Kidney Disease Drugs Market Report 2020
1 Chronic Kidney Disease Drugs Product Definition
2 Global Chronic Kidney Disease Drugs Market Manufacturer Share and Market Overview
3 Manufacturer Chronic Kidney Disease Drugs Business Introduction
4 Global Chronic Kidney Disease Drugs Market Segmentation (Region Level)
5 Global Chronic Kidney Disease Drugs Market Segmentation (Product Type Level)
6 Global Chronic Kidney Disease Drugs Market Segmentation (Industry Level)
7 Global Chronic Kidney Disease Drugs Market Segmentation (Channel Level)
8 Chronic Kidney Disease Drugs Market Forecast 2020-2025
9 Chronic Kidney Disease Drugs Segmentation Product Type
10 Chronic Kidney Disease Drugs Segmentation Industry………………. Request free sample to get a complete Table of Content
About us
BusinessindustryReports.com is digital database of comprehensive market reports for global industries. As a market research company, we take pride in equipping our clients with insights and data that holds the power to truly make a difference to their business. Our mission is singular and well-defined – we want to help our clients envisage their business environment so that they are able to make informed, strategic and therefore successful decisions for themselves.
Media Contact
Business Industry Reports
Pune – India
sales@businessindustryreports.com
+19376349940
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Chronic Kidney Disease Drugs Market Analysis Report: Reveals Lookout at Biggest Trends Hitting the Industry Over the Coming Years 2020-2025 here
News-ID: 2176327 • Views: …
More Releases from Business Industry Reports
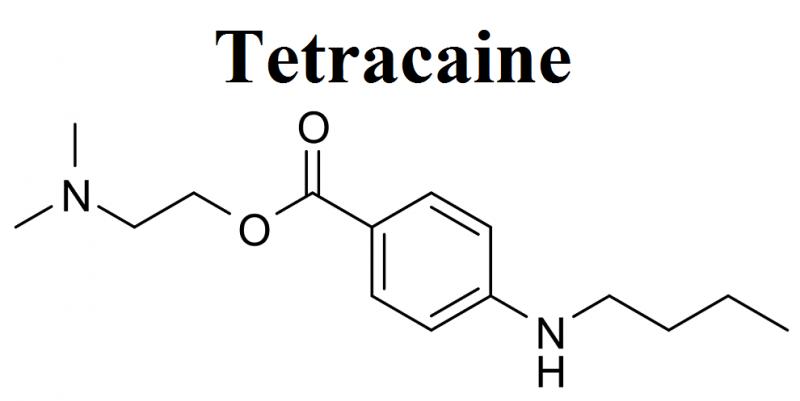
Tetracaine Market Exhibits a Lucrative Growth Potential during 2021-2025 | Endo …
BusinessIndustryReports has recently broadcasted a new study to its broad research portfolio, which is titled as “Global Tetracaine Market” Research Report 2021 provides an in-depth analysis of the Tetracaine with the forecast of market size and growth. The analysis includes addressable market, market by volume, market share by business type and by segment (external and in-house). The research study examines the Tetracaine on the basis of a number of criteria,…

Cyber Warfare Market Evenly Poised To Reach A Market Value of US$ By Share, Size …
Overview of Global Cyber Warfare Market:
This report provides in-depth study of “Global Cyber Warfare Market 2021” using SWOT analysis i.e. Strength, Weakness, Opportunities, and Threat to the organization. The Cyber Warfare Market report also provides an in-depth survey of key players in the market organization.
According to the market research study, the Cyber Warfare is virtual conflict between state, organization, or country by the use of computer technology to disrupt activities…
Covid-19 Impact on Cranio Maxillofacial Implant Market 2021-2025: Business Growt …
Overview of Global Cranio Maxillofacial Implant Market:
This report provides in-depth study of “Global Cranio Maxillofacial Implant Market 2021” using SWOT analysis i.e. Strength, Weakness, Opportunities, and Threat to the organization. The Cranio Maxillofacial Implant Market report also provides an in-depth survey of key players in the market organization.
According to the market research study, Craniomaxillofacial Implants are medical implants used in surgeries of maxillofacial region such as, head, face, neck, oral,…

Beginning of the bloom: The Rise of the Biohacking Market 2021-2025 | Global Key …
Global Biohacking Market Synopsis:
The report covers a forecast and an analysis of the Biohacking Market on a global and regional level. The study provides historical data for 2015, 2016, 2017 and 2018 along with a forecast from 2020 to 2025 based on revenue (USD Million) and volume (Kilotons). The study includes drivers and restraints of the Biohacking Market along with the impact they have on the demand over the forecast…
More Releases for Drug
Injectable Drug Delivery Market Injectable Drug Delivery Market
Leading market research firm SkyQuest Technology Group recently released a study titled ' Injectable Drug Delivery Market Global Size, Share, Growth, Industry Trends, Opportunity and Forecast 2024-2031,' This study Injectable Drug Delivery report offers a thorough analysis of the market, as well as competitor and geographical analysis and a focus on the most recent technological developments. The research study on the Injectable Drug Delivery Market extensively demonstrates existing and upcoming…
Global Advanced Drug Delivery Systems Market Size - By Product Type(Oral Drug De …
Market Overview and Report Coverage
Advanced Drug Delivery Systems (ADDS) refer to innovative technologies designed to improve the administration and efficacy of therapeutics, enhancing the way medications are delivered to targeted areas within the body. These systems aim to optimize treatment outcomes by increasing the bioavailability, reducing side effects, and facilitating controlled drug release. Employing methods such as nanoparticles, liposomes, and implantable pumps, ADDS are revolutionizing personalized medicine and expanding therapeutic…
Global Cancer Antibody Drug Conjugate Market Size, Drug Sales, Drug Dosage, Pric …
Global Cancer Antibody Drug Conjugate Market Size, Drug Sales, Drug Dosage, Price, and Clinical Trials Outlook 2029 Report Highlights:
* Global Antibody Drug Conjugates Market Opportunity: > 40 Billion By 2029
* Global and Regional Antibody Drug Conjugate Market Insight
* Approved Drugs Sales Insight Global and Regional, Yearly and Quarterly, 2019 -2023
* Approved Antibody Drug Conjugates - Availability, Dosage and Price Insight
* Insight On Antibody Drug Conjugates In Clinical Trials: > 550…
Alcohol Testing And Drug Testing Equipment Market 2025 Segmentation, Application …
Market Study Report, LLC, has compiled an exhaustive research study of the ‘Alcohol Testing And Drug Testing Equipment market’, detailing every single market driver and intricately analyzing the business vertical. This ‘Alcohol Testing And Drug Testing Equipment market’ study will aid in seeking out new business opportunities and fine-tuning existing marketing strategies through insights regarding SWOT analysis, market valuation, competitive spectrum, regional share, and revenue predictions.
Alcohol abuse and drug…
How much Diabetes Drug Market Impact Worldwide Medical Drug Industry?
Diabetes Drug Market From an insight perspective, the market report focuses on various levels of analyses — industry analysis, market rank analysis, and company profiles, which together comprise and discuss basic views on the competitive landscape, high-growth regions, and countries as well as their respective regulatory policies, Types ,Applications and opportunities in the market.
Diabetes is a metabolic disorder in which the body glucose level is elevated. There are two types of diabetes…
Hepatitis Drug Market Hepatitis Drug Clinical Pipeline Report 2023
For Report Sample Contact: neeraj@kuickresearch.com or +91-11-47067990
Report Table of Contents
1. Introduction to Hepatitis Disease
1.1 Prologue
1.1.1 History of Hepatitis
1.1.2 Causes of Hepatitis Disease
1.2 Types of Viruses which are Responsible for Hepatitis Disease
2. Global Prevalence of Hepatitis Infection
3. Available Drug Classes for Hepatitis Disease Treatment
3.1 Interferon Alfa Therapy
3.2 Protease Inhibitors Therapy
3.3 Polymerase…