Press release
Global Randomization and Trial Supply Management (RTSM) Market Analysis, Size,Share, Trend, Growth, Analysis and Forecast 2020 to 2027||Players Included-Dassault Systèmes, Parexel International Corporation., Bioclinica., DATATRAK Int., ERT Clinical, IBM
Data Bridge Market Research’s Latest Study on “Randomization and Trial Supply Management Market” report 2020-2027 provides an Overview of the current market situation, competitive analysis, product scope, market research, opportunities, driving force, and market risks. The report contains market forecast to 2027 related to market size, Consumption, gross margin, price, revenue, production, CAGR and other substantial factors. While emphasizing the key driving and restraining forces for this market, the report also offers a complete study of the future trends and developments of the market.The Randomization and Trial Supply Management market report provides a basic overview of the industry, including definitions, classifications, applications, and chain structures. Randomization and Trial Supply Management market analysis is provided to international markets, including development trends, competitive environment analysis, and key regions development status.
Get Sample Analysis of This Market Information: @ https://www.databridgemarketresearch.com/request-a-sample/?dbmr=global-randomization-and-trial-supply-management-rtsm-market
Randomization and trial supply management (RTSM) market is expected to gain market growth in the forecast period of 2020 to 2027. Data Bridge Market Research analyses the market to grow with a CAGR of 17.20% in the above-mentioned forecast period.
The major players covered in the randomization and trial supply management (RTSM) market report are Oracle, Dassault Systèmes, Parexel International Corporation., Bioclinica., DATATRAK Int., ERT Clinical, IBM Corporation, OmniComm Systems, Inc. MaxisIT, Bio-Optronics, Inc., eClinical Solutions LLC., among other domestic and global players. Market share data is available for global, North America, Europe, Asia-Pacific (APAC), Middle East and Africa (MEA) and South America separately. DBMR analysts understand competitive strengths and provide competitive analysis for each competitor separately.
Table of Content:
Part 01: Executive Summary
Part 02: Scope of the Report
Part 03: Research Methodology
Part 04: Randomization and Trial Supply Management Market Landscape
• Market Ecosystem
• Market Characteristics
• Market Segmentation Analysis
Part 05: Market Sizing
• Market Definition
• Market Sizing 2019
• Market Size And Forecast 2020-2027
Part 06: Customer Landscape
Part 07: Randomization and Trial Supply Management Market Regional Landscape
• Geographical Segmentation
• Regional Comparison
• Americas – Market Size And Forecast 2020-2027
• EMEA – Market Size And Forecast 2020-2027
• APAC – Market Size And Forecast 2020-2027
Part 08: Decision Framework
Part 09: Drivers And Challenges
• Market Drivers
• Market Challenges
Part 10: Randomization and Trial Supply Management Market Trends
Part 11: Vendor Landscape
• Overview
• Landscape Disruption
• Vendors Covered
• Vendor Classification
• Market Positioning Of Vendors
Get Full Table Of content @ https://www.databridgemarketresearch.com/toc/?dbmr=global-randomization-and-trial-supply-management-rtsm-market
Healthcare Infrastructure growth Installed base and New Technology Penetration
Randomization and trial supply management (RTSM) market also provides you with detailed market analysis for every country growth in healthcare expenditure for capital equipment, installed base of different kind of products for randomization and trial supply management (RTSM) market, impact of technology using life line curves and changes in healthcare regulatory scenarios and their impact on the randomization and trial supply management (RTSM) market. The data is available for historic period 2010 to 2018.
Segmentation:Global Randomization and Trial Supply Management (RTSM) Market
Randomization and Trial Supply Management (RTSM) Market By Delivery Mode
(Web-Hosted (On-Demand) Solutions, Licensed Enterprise (On-Premise) Solutions, Cloud-Based (SaaS) Solutions),
Randomization and Trial Supply Management (RTSM) Market By Clinical Trial Phase
(Phase I Clinical Trials, Phase II Clinical Trials, Phase III Clinical Trials, Phase IV Clinical Trials),
Randomization and Trial Supply Management (RTSM) Market By End User
(Pharmaceutical and Biopharmaceutical Companies, Contract Research Organizations, Consulting Service Companies, Medical Device Manufacturers, Hospitals, Academic Research Institutes),
Randomization and Trial Supply Management (RTSM) Market By Country
(U.S., Canada, Mexico, Germany, Italy, U.K., France, Spain, Netherland, Belgium, Switzerland, Turkey, Russia, Rest of Europe, Japan, China, India, South Korea, Australia, Singapore, Malaysia, Thailand, Indonesia, Philippines, Rest of Asia-Pacific, Brazil, Argentina, Rest of South America, South Africa, Saudi Arabia, UAE, Egypt, Israel, Rest of Middle East & Africa)
Want Full Report? Enquire Here @https://www.databridgemarketresearch.com/inquire-before-buying/?dbmr=global-randomization-and-trial-supply-management-rtsm-market
Contact:
Data Bridge Market Research
US: +1 888 387 2818
UK: +44 208 089 1725
Hong Kong: +852 8192 7475
Email: Corporatesales@databridgemarketresearch.com
About Us:
Data Bridge Market Research set forth itself as an unconventional and neoteric Market research and consulting firm with unparalleled level of resilience and integrated approaches. We are determined to unearth the best market opportunities and foster efficient information for your business to thrive in the market. Data Bridge Market Research provides appropriate solutions to the complex business challenges and initiates an effortless decision-making process.
Data Bridge adepts in creating satisfied clients who reckon upon our services and rely on our hard work with certitude. Get Customization and Discount on Report by emailing sopan.gedam@databridgemarketresearch.com . We are content with our glorious 99.9 % client satisfying rate.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global Randomization and Trial Supply Management (RTSM) Market Analysis, Size,Share, Trend, Growth, Analysis and Forecast 2020 to 2027||Players Included-Dassault Systèmes, Parexel International Corporation., Bioclinica., DATATRAK Int., ERT Clinical, IBM here
News-ID: 2140833 • Views: …
More Releases from Data Bridge Market Research

Scented Candle Market Shows Strong Growth Driven by Wellness and Home Décor Tr …
The global scented candle market is on track for significant expansion, increasing from an estimated USD 3.60 billion in 2024 to USD 6.00 billion by 2032, registering a strong CAGR of 6.60%. Rising consumer interest in home ambiance, wellness, and premium lifestyle products continues to drive market demand.
Get More Detail: https://www.databridgemarketresearch.com/reports/global-scented-candle-market
Market Growth Drivers
The scented candle market has evolved beyond being just a decorative item. Key growth factors include:
Home Fragrance &…
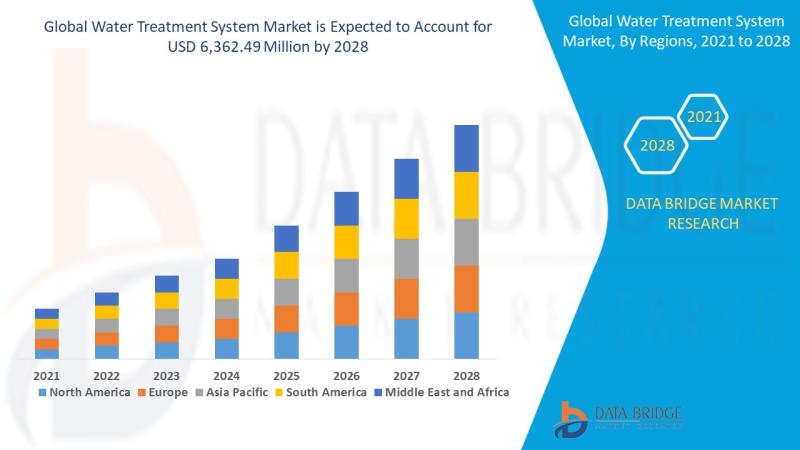
Water Treatment System Market: Sustaining the Future of Clean Water
Introduction
Understanding Water Treatment Systems
Water treatment systems are designed to purify and disinfect water for various uses-drinking, industrial processes, irrigation, and wastewater reuse. These systems eliminate contaminants such as bacteria, viruses, heavy metals, chemicals, and particulates, making water safe and sustainable for consumption and use.
Importance in Global Sustainability
Clean water is essential to life and industrial progress. With growing water demand and pollution, water treatment systems are now critical infrastructure across the…

Veterinary X-Ray Market Size, Analysis, Scope, Demand, Opportunities, Statistics
According to Data Bridge Market Research The global Veterinary X-Ray market size was valued at USD 915.19 million in 2024 and is projected to reach USD 1576.00 million by 2032, with a CAGR of 7.03 % during the forecast period of 2025 to 2032.
With increasing globalization and digital disruption, the Equine X-Ray Solutions Market is expanding across multiple industries, . Market research data indicates that businesses in the Companion Animal…

Veterinary X-Ray Market Size, Analysis, Scope, Demand, Opportunities, Statistics
According to Data Bridge Market Research The global Veterinary X-Ray market size was valued at USD 915.19 million in 2024 and is projected to reach USD 1576.00 million by 2032, with a CAGR of 7.03 % during the forecast period of 2025 to 2032.
With increasing globalization and digital disruption, the Equine X-Ray Solutions Market is expanding across multiple industries, . Market research data indicates that businesses in the Companion Animal…
More Releases for Trial
Clinical Trial Investigative Site Network Market Clinical Trial Investigative Si …
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Clinical Trial Investigative Site Network Market - (By Therapeutic Areas (Oncology, Cardiology, CNS, Pain Management, Endocrine, Others), By Phase (Phase I, Phase II, Phase III, Phase IV), By End-use (Sponsor, CRO)), Trends, Industry Competition Analysis, Revenue and Forecast To 2034."
According to the latest research by InsightAce Analytic, the Global Clinical Trial Investigative Site Network Market…
Transformative Trends Impacting the Electronic Trial Master File (eTMF) Systems …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Electronic Trial Master File (eTMF) Systems Market Size By 2025?
The market size of the electronic trial master file (eTMF) systems has experienced fast growth over recent years. The market is projected to increase from $1.36 billion in 2024 to $1.55 billion in 2025, with…
Transformative Trends Impacting the Electronic Trial Master File (eTMF) Systems …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Electronic Trial Master File (eTMF) Systems Market Size By 2025?
The market size of the electronic trial master file (eTMF) systems has experienced fast growth over recent years. The market is projected to increase from $1.36 billion in 2024 to $1.55 billion in 2025, with…
Clinical Trial Imaging market
The Clinical Trial Imaging market crossed the US$ 1.09 billion mark in 2022 and is expected to hit US$ 1.94 billion by 2030, recording a CAGR of 7.5% during the forecast period.
Rising R&D spending, a rapidly growing pharmaceutical industry, and an increase in the number of contract research organizations are some of the major factors driving the market's growth. There has been an increase in pharmaceutical companies due to the…
Clinical Trial Logistics
Clinical Trial Logistics
16th to 17th May 2011, Marriott Regents Park, London, United Kingdom.
It currently costs just over £500 million ($800 million) to bring a new chemical to market and development timelines continue to fall in the 10-15 year range. A key reason for high R&D costs is due to logistical failures including failure to recruit patients on time. A way to avoid this is to move clinical trials…
Clinical Trial Logistics
Announcing SMi's 5th annual…
Clinical Trial Logistics conference
16th and 17th May 2011, Central London, UK
www.smi-online.co.uk/2011logistics-london6.asp
It currently costs just over £500 million ($800 million) to bring a new chemical to market and development timelines continue to fall in the 10-15 year range. A key reason for high R&D costs is due to logistical failures including failure to recruit patients on time. A way to avoid this is to move clinical…
