Press release
Global Gene Therapy Market is forecast to hit $363 million by 2022
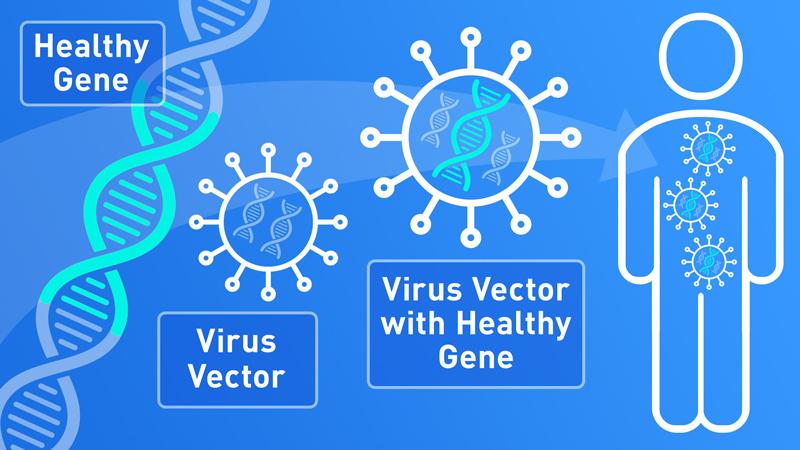
Global gene therapy market is forecast to hit $363 million by 2022 from $x million in 2017. Strengthened by recent approvals of Kymriah, Yescarta and Luxturna in the US, and a committed European, Japanese and Chinese environment, gene therapy is set to become a significant player in the bio-pharmaceutical industry. The space covers many therapeutic areas specifically, oncology, rare diseases, Parkinson's, HIV, severe combined immuno-deficiencies (SCID) and hemophilia. Recently, in November 2017, the FDA indicated that gene therapies will now qualify for a fast approval process, which will bring more therapies to market faster. However, the space also has significant challenges, such as manufacturing logistics, reimbursement and its high cost. This 310 page market analysis cutting-edge report tackles this growing but challenging industry, it highlights its strengths, weaknesses and opportunities and provides a comprehensive account of major companies, clinical trials and technological advancement.Browse Complete Global Gene Therapy Market Analysis and Forecasts Report at https://www.reportsnreports.com/reports/1370259-global-gene-therapy-market-analysis-forecast-to-2022.html
Since the FDA approved Kymriah (tisagenlecleucel), Yescarta (Axicabtagene ciloleucel) and Luxturna (voretigene neparvovec-rzyl) in 2017, the US gene therapy space has expanded significantly, underlined by the fact that over 55% of completed and ongoing trials are located in this geographic. Growth in the gene therapy industry has resulted in new commercial initiatives and the emergence of new startups and spin-off biotechs. Furthermore, gene therapy specifically has raised well over $600 million of venture capital in the last five years. Early stage companies have raised seed, Series A and Series B investment steadily since the market took off, including Spark Therapeutics, Avalanche Biotech, uniQure, Voyager Therapeutics, Editas Medicine and GenSight.
In 2017, the Gene Therapy Market for technologies, services and products was estimated to be worth $x million, with a potential to reach $363 million by 2022. The main market space is cancer which currently holds x% market share. This indication generated $x million in 2017 and will generate $x million in 2022. This is followed by rare diseases, cardiovascular, neurological and ocular indications. Looking at the market by technology, at present, gene product therapeutics generate the majority of revenue with over $x million in 2017, growing to $x million by 2022. Viral vectors are set to generate $x million in 2017, and will rise to $x million in 2022, and by then gene therapy services such as vector development and transfection will hit $x million. At present, the Americas have penetrated the market significantly with 65% geographic share, followed by Europe (x%) and the RoW (x%).
Get Free Sample Copy of Global Gene Therapy Market Research Report at https://www.reportsnreports.com/contacts/requestsample.aspx?name=1370259
Gene therapy products approved between the years 2003 and 2017 include Gendicine, Oncorine, Rexin-G, Neovasculgen, Glybera, Imlygic, Strimvelis, Zalmoxis, Kymriah, Yescarta and Luxturna. Gendicine was approved for head and neck squamous cell carcinoma and has been in the Chinese market since 2003. Rexin-G was approved in the Philippines back in 2007 for the treatment of primary and metastatic cancer. Oncorine was approved in China in 2005 for nasopharyngeal carcinoma. The Russian market has Neovasculgen from 2011 for the treatment of peripheral arterial disease (PAD) and critical limb ischemia. The first gene therapy approved in E.U. was Glybera in 2012 for the treatment of familial lipoprotein lipase deficiency (LPL), however in October 2017 it was pulled from the market due to lack of patient demand. In 2015, Imlygic was approved in E.U. and also in the U.S. to treat melanoma, and Phase II results released in 2017 indicated its efficacy in combination with the checkpoint-inhibitor, Yervoy. In the E.U., Strimvelis was approved in 2016 for the treatment of adenosine deaminase severe combined immunodeficiency (ADA-SCID). In 2016, Zalmoxis was approved in E.U. for the treatment of leukemia. 2017 was a bumper year for gene therapy with Kymriah, Yescarta and Luxturna all gaining FDA approval.
Renewed interest has encouraged start-up companies to affiliate with academic centers for tech know-how. As clinical trials advance towards licensure, more meticulous product characterization using improved analytical methods and progressively higher regulatory compliance will be required. Some of the ongoing clinical trials are closing on to produce promising results, including one for hemophilia B caused by the deficiency of Factor IX using a recombinant adeno-associated virus (AAV) as a vector. The product candidate if succeeds will be a relatively cheaper alternative to the expensive and lifelong factor replacement therapy.
A second example of a successful outcome in gene therapy are studies conducted by independent laboratories focusing on sub-retinal delivery of recombinant AAV expressing retinal pigment epithelial RPE65 for Leber Congenital Amaurosis Type 2. A third example is the clinical trial involving nine children with X-linked severe combined immunodeficiency (SCID-X1) treated with autologous bone marrow CD34+ cells transduced with a self-inactivating (SIN) ?-retroviral vector expressing the IL-2 receptor ?-chain. This novel strategy involves ex vivo gene transfer using recombinant retroviral or lentiviral vectors of chimeric antigen receptors consisting of antibody-binding domains fused to T-cell-signaling domains into patient T lymphocytes.
Access this Research Report at https://www.reportsnreports.com/contacts/discount.aspx?name=1370259
As gene therapies are generally meant for one time or short duration treatments, they are customized to individuals confined to small patient populations. Therefore, manufacturing firms are expected to seek premium prices for these therapies. Because of this, these therapies will have to face valuation and reimbursement challenges. Stakeholders will show reservations about the hefty price tags and they will require significant data to be convinced. With the removal of Gylbera from the EU market in 2017, due to the fact that only one patient was treated with the drug, all eyes are focused on the number of end patients that will be treated, and their ability to pay. To that end, launching of new drugs may have to be delayed in order to collect more data for payers. Furthermore, annuity based reimbursement agreements and pay-for-performance scenarios will have to be tackled.
This report provides the reader with:
o Current Global Market Worth and Forecast with CAGR Through 2022
o Sub-Market Worth by Therapeutic Area (Cancer, Rare Diseases, Cardiovascular, Neurological, Ocular) and Forecast with CAGR Through 2022
o Sub-Market Worth by Geography (Americas, Europe, RoW) and Forecast with CAGR Through 2022
o Sub-Market Worth by Technology (Gene Product, Service, Viral Vectors) and Forecast with CAGR Through 2022
o Insight into gene therapy technologies, challenges associated with developing therapeutic genes and disadvantages of gene therapy.
o Full outline of the gene therapy industry from the formative years through to products discovered during 1990 and 2017.
o Detailed descriptions of commercialized products approved between 2003 and 2017 that include: Gendicine, Rexin-G, Oncorine, Neovasculgen, Glybera, Imlygic, Strimvelis, Zalmoxis, Kymriah, Yescarta and Luxturna.
o Description of seven of the Phase III product candidates that include: Generx, Collategene, LentiGlobin, Lenti-D, VM-202, Invosa and GS-010.
o Description of 21 Phase II product candidates that are set to have significant market share.
o Commercialization status of gene therapies in by geographic region
o Evaluation of gene therapy pricing
o Description of the firstever warranty offer by GSK for Strimvelis.
o A detailed analysis of various types of viruses used as vectors.
o Description of clinical applications of gene therapy and the various genetic and infectious diseases addressed by gene therapy.
o Description of 77 companies that are directly and indirectly associated with gene therapy industry.
Get Now Global Gene Therapy Market Research Report at https://www.reportsnreports.com/purchase.aspx?name=1370259
Key Questions Answered in this Report:
o What is the size of gene therapy market?
o What is the CAGR and market size over the next five years?
o What are the different sub-markets and their worth/CAGR over the next five years?
o What is gene augmentation therapy?
o What is suicide gene therapy?
o How is ex vivo gene delivery different from in vivo gene delivery?
o What are the types of gene therapies classified on the basis of targeted cell types?
o What is the role of CRISPR technology in gene therapy?
o What are the approved gene therapy products?
o How many gene therapy product candidates have reached the Phase III stage?
o How many Phase II gene therapy product candidates are there?
o What is the commercialization status of gene therapies in E.U. member countries?
o What are the prices of gene therapy products?
o What are the reasons for this extortionate pricing of gene therapies?
o Which company is offering warranty for its gene- therapy product?
o What is the current strength of gene therapy industry?
o Is it true that the real strength of gene therapy industry is based on the number of clinical trials?
o What is the total number of ongoing clinical trials as of 2017?
o What is the distribution of clinical trials by geography?
o Which countries are associated with gene therapy clinical trials?
o What are the major indications addressed by the clinical trials?
o Which genes are transferred in these clinical trials?
o How many Big Pharma are associated with the gene therapy industry?
o What are non-viral and viral vectors?
o What are the various features of viral vectors?
o Which viral vectors are predominantly used in gene therapy clinical trials?
o What are the major diseases addressed by therapeutic genes?
o Where is the gene therapy market heading, and what opportunities and challenges will it face?
ReportsnReports.com is your single source for all market research needs. Our database includes 500,000+ market research reports from over 95 leading global publishers & in-depth market research studies of over 5000 micro markets. With comprehensive information about the publishers and the industries for which they publish market research reports, we help you in your purchase decision by mapping your information needs with our huge collection of reports.
+ 1 888 391 5441
sales@reportsandreports.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global Gene Therapy Market is forecast to hit $363 million by 2022 here
News-ID: 1947998 • Views: …
More Releases from ReportsnReports
DeviceCon Series 2024 - UK Edition | MarketsandMarkets
Future Forward: Redefining Healthcare with Cutting-Edge Devices
Welcome to DeviceCon Series 2024 - Where Innovation Meets Impact!
Join us on March 21-22 at Millennium Gloucester Hotel, 4-18 Harrington Gardens, London SW7 4LH for a groundbreaking convergence of knowledge, ideas, and technology. MarketsandMarkets proudly presents the DeviceCon Series, an extraordinary blend of four conferences that promise to redefine the landscape of innovation in medical and diagnostic devices.
Register Now @ https://events.marketsandmarkets.com/devicecon-series-uk-edition-2024/register
MarketsandMarkets presents…

5th Annual MarketsandMarkets Infectious Disease and Molecular Diagnostics Confer …
London, March 7, 2024 - MarketsandMarkets is thrilled to announce the eagerly awaited 5th Annual Infectious Disease and Molecular Diagnostics Conference, scheduled to take place on March 21st - 22nd, 2024, at the prestigious Millennium Gloucester Hotel, located at 4-18 Harrington Gardens, London SW7 4LH.
This conference promises to be a groundbreaking event, showcasing the latest trends and insights in diagnosis, as well as unveiling cutting-edge technologies that are revolutionizing the…

Infection Control, Sterilization & Decontamination Conference |21st - 22nd March …
MarketsandMarkets is pleased to announce its 8th Annual Infection Control, Sterilisation, and Decontamination in Healthcare Conference, which will take place March 21-22, 2024, in London, UK. With the increased risk of infection due to improper sterilisation and decontamination practices, the safety of patients and healthcare workers is of paramount importance nowadays.
Enquire Now @ https://events.marketsandmarkets.com/infection-control-sterilization-and-decontamination-conference/
This conference aims to bring together all the stakeholders to discuss the obstacles in achieving…

Breast Augmentation Market Key Players, Demands, Cost, Size, Procedure, Shape, S …
The global Breast Augmentation Market in terms of revenue was estimated to be worth $900 million in 2020 and is poised to reach $1,692 million by 2025, growing at a CAGR of 13.4% from 2020 to 2025. The new research study consists of an industry trend analysis of the market. The new research study consists of industry trends, pricing analysis, patent analysis, conference and webinar materials, key stakeholders, and buying…
More Releases for Gene
DNA and Gene Cloning Services Market Expands with Growing Focus on Complex Gene …
InsightAce Analytic Pvt. Ltd. has announced the publication of a market research report titled "Global DNA and Gene Cloning Services Market by Type of Service Offered (Custom Cloning, Sub-cloning, Gene Synthesis, and Others), Type of Gene (Complex Gene, Standard Gene, and Others), End-User Industry (Pharmaceutical, Academic and R&D, and Biotechnology Companies, and Others)- Market Outlook and Industry Analysis 2034"
The DNA and Gene Cloning Services Market Size is valued…
Evolving Market Trends In The CRISPR Gene Editing Industry: Innovative Gene Ther …
The CRISPR Gene Editing Market Report by The Business Research Company delivers a detailed market assessment, covering size projections from 2025 to 2034. This report explores crucial market trends, major drivers and market segmentation by [key segment categories].
What Is the Expected CRISPR Gene Editing Market Size During the Forecast Period?
The CRISPR gene editing market has grown exponentially in recent years. It will grow from $2.26 billion in 2024 to $2.88…
Cell and Gene Therapy Market Global Analysis By Type (Cell Therapy, Gene Therapy …
Gene and cell therapy uses genes and cells for the treatment of genetic diseases. Genetic diseases are caused by mutations, or errors in genes which can be passed down from one generation to another. Gene therapy aims to treat diseases by using genetic material, or DNA, to manipulate a patient's cells by replacing, changing or introducing genome into cells- either internally or externally. Cell therapy aims to treat diseases by…
Competitive and Opportunities Analysis of Gene Therapy Market of Gene Therapy Ma …
Global Gene Therapy Market accounted for US$ 2.05 billion in 2020 and is estimated to be US$ 12.29 billion by 2030 and is anticipated to register a CAGR of 19.8%. Gene therapy means fixing a working gene to an individual who features a damaged gene. The European Commission has approved this method for one particular treatment. The treatment by the merchandise Glybera uses an epidemic to infect muscle cells with…
Genetic Testing Market Size by Growth Opportunities, Top Key Players: GeneDx, In …
Genetic Testing Market Report provides an in-depth analysis of the overall market, The ripple effect of Coronavirus-Covid19 on the market needs to become part of strategy discussions to emerge strong. The report focuses on major key players, production details, their application, countries and also analyzes the global and key regions market potential and advantage, opportunity, and challenge, restraints, and risks.
The Top players Covered in report are GeneDx, Invitae, Pathway Genomics,…
Gene Synthesis Market by Top Manufacturers - Gene script, Gene Art (Thermofische …
The "Gene Synthesis Market" report Added by "Big Market Research", enumerates information about the industry in terms of market share, market size, revenue forecasts, and regional outlook. The report further illustrates competitive insights of key players in the business vertical followed by an overview of their diverse portfolios and growth strategies.
In the Gene Synthesis Market 2018 research report professionals describe the different facets of the industry with a specific goal…