Press release
Yoritex Inc. successfully completes comprehensive testing of its Medical Network HUB® prototype
Toronto, Ontario. – September 28, 2011. - Yoritex Inc., developer of the first medical diagnostics accessories for BlackBerry® smartphones, today announced the successful conclusion of its Medical Network HUB® prototype testing. Having successfully completed comprehensive hardware and software testing, Yoritex Inc. will commence manufacture of a pilot batch of Medical Network HUB® in October that accelerates development of its medical plug-in units line.During the eight-week comprehensive test Yoritex Inc. has schematically validated and re-evaluated the Medical Network HUB® prototype and validated the software architecture in the field. All modules, components and business processes of the Medical Network HUB® system were verified and assessed according to system functionality, non-functional and business requirements.
The testing results confirmed the functionality of the device platform. The core of the device is based on the new Freescale medical MCU. During testing, the USB connectivity between Medical Network HUB® and BlackBerry smartphone was verified.
Testers identified a need to enhance the device’s power management, leading Yoritex Inc. to enter into an agreement with Dialog Semiconductor, a provider of highly integrated innovative power management ICs. Dialog Semiconductor’s power management IC offers an energy-optimized solution suitable for portable handheld applications and facilitates the improvement of the technical characteristics of the Medical Network HUB® by Yoritex Inc. Yoritex Inc. and Dialog Semiconductor will now sign a long-term co-operation agreement.
Based on the positive test results, which are within approved according to the company test protocol, Yoritex Inc. is excited to move to the next stage of development. The company is now completing the developing of a medical front-end sub-system, which includes a set of measurement units for direct contact with patients’ bodies. The R&D stage of the newly developed ECG plug-in unit is now complete and design documentation available for manufacturing. Yoritex Inc. is also finalizing the development of major plug-in units that support blood pressure measurement, blood analysis, body temperature measurement and pulse oximetry.
Yoritex Inc. Shareholders have invested significantly in the development of the first mobile medical diagnostic device for Blackberry smartphones, opening new opportunities for its owners. The company has reached spectacular results, and is now inviting investors to place strategic investment that will allow Yoritex Inc. to begin the CE and FDA, certification process as well as commencing the commercialization of its products.
Leon Dain, President and CEO of Yoritex Inc., commented, “Our main goal is to make the Medical Network HUB® premiere in November”.
About Medical Network HUB®
Medical Network HUB® by Yoritex Inc. is the first USB-connected hardware accessory for BlackBerry smartphones, which enables over 50 different medical and non-medical measurements via plug-in units by Yoritex Inc. It provides full mHealth service on real-time transmit data basis.
Medical Network HUB® by Yoritex Inc. solution includes:
• Set of plug-in units for medical and non-medical parameter measurements;
• Independent, self-powered data processing system.
About Yoritex Inc.
Yoritex Inc. (www.yoritex.com) is a private held engineering research & development company located in Toronto, Canada. Its aim is to develop and manufacture mobile medical diagnostic devices. Yoritex' flagship product is the Medical Network HUB®, which is placed on the top of the medical diagnostic accessory product range by Yoritex Inc. for BlackBerry smartphones. From 2010 Yoritex Inc. is a Select Member of the BlackBerry® Alliance Program.
For more information, please visit http://www.yoritex.com/#/how-it-works-42/ or contact Yoritex Inc. at nda@yoritex.com
To view all press releases, please visit http://yoritex.com/#/companynews/
10 Mintwood Road,
Vaughan, ON L4J 9C3.
Canada
Phone: +1 416 8364662
Media Contact:
Nataly Dain,
Yoritex Inc. Media Director
Phone (in EU): +371 220 65135
Email nda@yoritex.com
www.yoritex.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Yoritex Inc. successfully completes comprehensive testing of its Medical Network HUB® prototype here
News-ID: 193927 • Views: …
More Releases from Yoritex Inc.
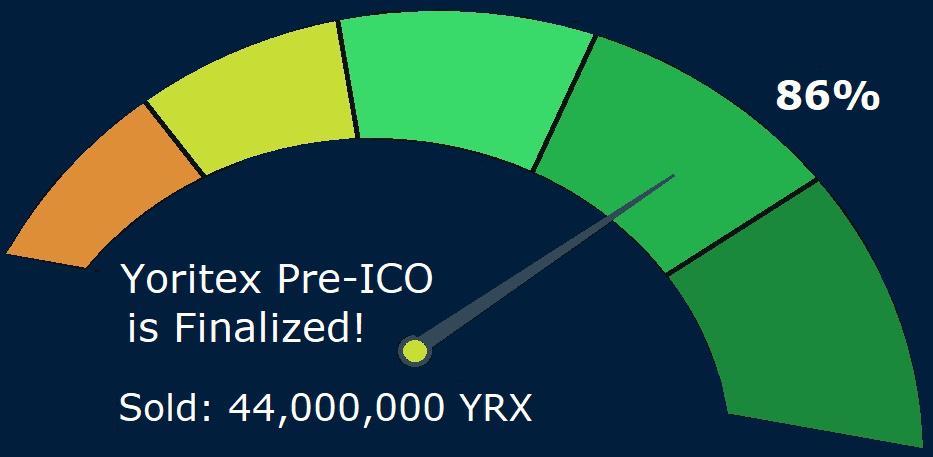
Yoritex Inc. Finalized Its Pre-ICO With Great Success
Company finished internal audit of early stage sale YRX distribution
The Toronto-based Fintech company Yoritex Inc., currently running the ICO campaign to support the development of its SimcoePay™, the First European Cross-Border Multicurrency Online Bank for EU Non-Residents, is pleased to announce that its expansion plans are well underway as it has finalized its Pre-ICO with great success.
44,000,000 YRX tokens were sold during the Pre-ICO, representing 86% of the…

Yoritex Inc. Offers 1000 EUR MasterCard Gift Cards to Loyal Buyers of YRX Tokens …
The Toronto-based Fintech company Yoritex Inc., is currently running the ICO campaign to support the development of its SimcoePay™, the First European Cross-Border Multicurrency Online Bank for EU Non-Residents, has announced initiation of the first Loyalty Program for all ICO participants.
Starting February 15, 2018 and till March 01, 2018 owners of YRX tokens can exchange their YRX tokens to the Limited Edition SimcoePay MasterCard Prepaid Gift Cards fully…

Yoritex is conducting a unique Initial Coin Offering campaign “1+90”
November 29th 2017, Toronto, Canada. - Canadian FinTech Company, Yoritex Inc., is proud to introduce SimcoePay, the first European cross-border multicurrency online bank for both residents and non-residents of Europe.
SimcoePay is officially announcing the start of its unique Initial Coin Offering (ICO) starting November 29, 2017 through to the 1st May 2018.
The campaign has received accolades from experts in the field of financial technologies and crypto-currencies which will be…
Yoritex Inc. have for the first time demonstrated the measuring possibilities of …
Toronto, Ontario. – December 29, 2011. - Yoritex Inc., developer of the USB medical diagnostics accessories for BlackBerry® smartphones, have demonstrated its Medical Network HUB® (YoriMed®).
Presentation shows functionality of YoriMed medical interface with Medical EСG Module by Freescale Semiconductor, connected to the BlackBerry Bold 9780 via micro USB. The presentation is available on YouTube from 27th of December 2011 (http://www.youtube.com/watch?v=u0raQ0rmL2I).
YoriMed® - is an evolutionary mHealth solution for Blackberry and…
More Releases for Medical
ECG Analysis System Market Research Report 2022 - GE Medical, Medical Econet, Gr …
A recent market research report added to repository of MR Accuracy Reports is an in-depth analysis of global ECG Analysis System. On the basis of historic growth analysis and current scenario of ECG Analysis System place, the report intends to offer actionable insights on global market growth projections. Authenticated data presented in report is based on findings of extensive primary and secondary research. Insights drawn from data serve as excellent…
ECG Analysis System Market Outlook: 2020 The Year On A Positive Note | GE Medica …
DataIntelo report titled Global ECG Analysis System Market provides detailed information and overview about the key influential factors required to make well informed business decision. This is a latest report, covering the current COVID-19 impact on the market. The pandemic of Coronavirus (COVID-19) has affected every aspect of life globally. This has brought along several changes in market conditions. The rapidly changing market scenario and initial and future assessment of…
Medical Tracheostomy Tube Market Overall Study Report 2020-2027 | Players Medtro …
The latest report added by Stratagem Market Insights gives deep insights into the drivers and restraints in the Worldwide Medical Tracheostomy Tube Market. The research report "Global Medical Tracheostomy Tube Market Size and Growth Forecast to 2027" provide a comprehensive take on the overall market. Analysts have carefully evaluated the milestones achieved by the global Medical Tracheostomy Tube market and the current trends that are likely to shape its future.…
Global Embolization Particle Market 2019 - Sirtex Medical, Merit Medical, Cook M …
The global "Embolization Particle Market" report delivers a comprehensive and systematic framework of the Embolization Particle market at a global level that includes all the key aspects related to it. The data is collected from different sources allied to the global Embolization Particle market and the research team meticulously analyze the gathered data with the help of various analytical tools and present their opinion based on analysis and calculations. The…
Medical Casting & Splinting Market 2019: Top Key players are 3M, DJO Global, BSN …
Medical Casting & Splinting Market 2019 Report analyses the industry status, size, share, trends, growth opportunity, competition landscape and forecast to 2025. This report also provides data on patterns, improvements, target business sectors, limits and advancements. Furthermore, this research report categorizes the market by companies, region, type and end-use industry.
Get Sample Copy of this Report@ https://www.researchreportsworld.com/enquiry/request-sample/13718924
Global Medical Casting & Splinting market 2019 research provides a basic overview of…
Global Oxygen Pressure Regulator Market 2017 : Precision Medical, Smiths Medical …
Oxygen Pressure Regulator
A market study based on the " Oxygen Pressure Regulator Market " across the globe, recently added to the repository of Market Research, is titled ‘Global Oxygen Pressure Regulator Market 2017’. The research report analyses the historical as well as present performance of the worldwide Oxygen Pressure Regulator industry, and makes predictions on the future status of Oxygen Pressure Regulator market on the basis of this analysis.
Get Free…