Press release
Kanban Zone Moves to Free Trial Mode
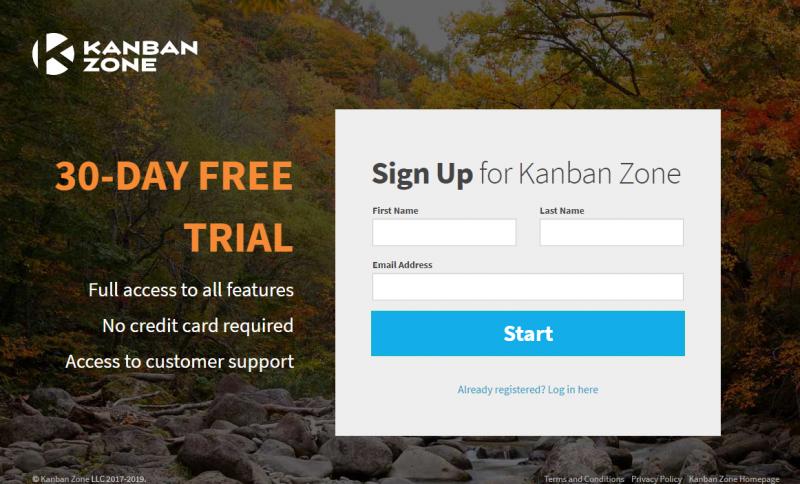
Kanban Zone today announced the move to a Free Trial model, a new, significantly improved experience for new subscribers and first-time users. The Free Trial model, offered upon Sign Up, is available for 30 days to all new users, no credit card required. It includes all features, which offers is a significantly upgraded experience to new users than the limited Free plan. There are also improvements for existing users when creating a new board.“We want to provide our users a comprehensive, yet easy-to-use tool that will help them achieve their goals more efficiently. We believe that adding the 30-Day Free Trial model will support this by allowing new subscribers and first-time users to take the time to get to know the tool (Kanban Zone), familiarize themselves with the Kanban method, and explore all Kanban Zone features, basic and advanced, and receive support and guidance in setting up their boards and using the full potential of Kanban Zone.” – says Dimitri Ponomareff, co-founder of Kanban Zone.
The main difference between the Free Plan and the Free Trial model is that the Free Trial model gives new users unrestricted access to all features for 30 days. The 3 existing plans: Personal, Start-Up and Enterprise are still available and have not changed. At any time during the Free Trial new users can select one of these 3 plans. The Personal plan is still free.
Kanban Zone, the most advanced online Kanban tool on the market, hopes that adding the Free Trial model can help teams and individuals to significantly improve the way they work, communicate and get work done. They continually work on improving the product and offering outstanding customer support.
The Free Trial rolled out on 20 November 2019. Now, as a holiday special, new subscribers can sign up for an extended 60-day Free Trial on the following link https://lnkd.in/g57MyFh.
Kanban Zone
7047 E. Greenway Parkway, Suite 250
Scottsdale, AZ 85254, USA
Press Contact:
Ivana Sarandeska
social@kanbanzone.com
+1 480-269-0590
Kanban Zone was founded by two Agile coaches, seeking the best online Kanban tool for their clients. They built Kanban Zone based on their Kanban and Agile expertise. The tool provides the most effective online Kanban tool on the market, with the most advanced Kanban board editor and specialized templates to help you get started. The co-founders and constantly seek better ways to share, teach and implement new ways of working in all environments, and as a result, they constantly work on ways to improve Kanban Zone.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Kanban Zone Moves to Free Trial Mode here
News-ID: 1878806 • Views: …
More Releases from Kanban Zone

Kanban Zone Partners with Modus Institute to Deliver Expert-Led Services That He …
New strategic services partnership brings world-class coaching, Kanban system design, and visual work expertise to Kanban Zone customers.
Scottsdale, AZ - August 19, 2025 - Kanban Zone, a leading visual work management platform, today announced a strategic partnership with Modus Institute, the global experts in visual collaboration and creators of Personal Kanban, Lean Coffee, and The Right Environment. This partnership enables organizations to not only visualize their work through software-but to…

Kanban Zone Wins the Winter 2024 Top Performer Award in Kanban Software from Sou …
Kanban Zone is proud to be a winner of the Top Performer award from SourceForge, the world's largest software reviews and comparison website.
Scottsdale, Arizona, February 09, 2024 - Kanban Zone, the top-rated visual planning software, today announced that it has been awarded a Winter 2024 Top Performer Award by SourceForge, the world's largest software and services review and comparison website. This award recognizes exceptional companies and products with a significant…

Kanban Zone Launches a Trello Integration
The New Integration Enables Trello Users to Import All Their Data into Kanban Zone Seamlessly
Scottsdale, Arizona, June 29, 2023 - Today, Kanban Zone announced the launch of a seamless integration between Trello and Kanban Zone.
With this new feature, users can import all of their Trello data, including cards, labels, comments, and more with just a couple of clicks.
The new integration makes it easy to move all types of Trello…

Kanban Zone Launches Performance Tuning Metrics to Help Improve Productivity
Scottsdale, Arizona, April 1, 2023 -- Kanban Zone today announced the launch of Performance Tuning Metrics to help users track their work better and improve their processes. Using the Performance Tuning Metrics, users will be able to see detailed analytics and reports, such as the average time spent and/or blocked in each column, Flow Efficiency, Throughput, Lead Time, and Cycle Time. This data can then be used to identify areas…
More Releases for Trial
Clinical Trial Investigative Site Network Market Clinical Trial Investigative Si …
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Clinical Trial Investigative Site Network Market - (By Therapeutic Areas (Oncology, Cardiology, CNS, Pain Management, Endocrine, Others), By Phase (Phase I, Phase II, Phase III, Phase IV), By End-use (Sponsor, CRO)), Trends, Industry Competition Analysis, Revenue and Forecast To 2034."
According to the latest research by InsightAce Analytic, the Global Clinical Trial Investigative Site Network Market…
Transformative Trends Impacting the Electronic Trial Master File (eTMF) Systems …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Electronic Trial Master File (eTMF) Systems Market Size By 2025?
The market size of the electronic trial master file (eTMF) systems has experienced fast growth over recent years. The market is projected to increase from $1.36 billion in 2024 to $1.55 billion in 2025, with…
Transformative Trends Impacting the Electronic Trial Master File (eTMF) Systems …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Electronic Trial Master File (eTMF) Systems Market Size By 2025?
The market size of the electronic trial master file (eTMF) systems has experienced fast growth over recent years. The market is projected to increase from $1.36 billion in 2024 to $1.55 billion in 2025, with…
Clinical Trial Management System
According to a new market report published by Persistence Market Research “Global Market Study on Clinical Trial Management System: Asia to Witness Highest Growth by 2019” the global clinical trial management system market was valued at USD 844.0 million in 2013 and is expected to grow at a CAGR of 14% from 2014 to 2019, to reach an estimated value of USD 1,848.5 million in 2019.
Request Report TOC @ https://www.persistencemarketresearch.com/methodology/3017
…
Clinical Trial Logistics
Clinical Trial Logistics
16th to 17th May 2011, Marriott Regents Park, London, United Kingdom.
It currently costs just over £500 million ($800 million) to bring a new chemical to market and development timelines continue to fall in the 10-15 year range. A key reason for high R&D costs is due to logistical failures including failure to recruit patients on time. A way to avoid this is to move clinical trials…
Clinical Trial Logistics
Announcing SMi's 5th annual…
Clinical Trial Logistics conference
16th and 17th May 2011, Central London, UK
www.smi-online.co.uk/2011logistics-london6.asp
It currently costs just over £500 million ($800 million) to bring a new chemical to market and development timelines continue to fall in the 10-15 year range. A key reason for high R&D costs is due to logistical failures including failure to recruit patients on time. A way to avoid this is to move clinical…