Press release
Global Orphan Drugs Market will multiply at impressive CAGR of 11.9% by 2024 & Top Key Players are Novartis AG, F. Hoffmann-La Roche Ltd, Celgene, Bristol-Myers Squibb Company, Shire pharmaceuticals and Pfizer
Orphan Drugs Market is estimated to witness a CAGR of 11.9% during the forecast period 2017–2023Orphan Drugs Market – Global Drivers, Restraints, Opportunities, Trends, and Forecasts: 2017–2023
Overview: Orphan diseases or rare diseases rarely occur among the people, which is nearly 7 out of 10,000 individuals. Around 5,000 to 8,000 rare diseases have been recognized and the origin of approximately 80% of these diseases is from the genes. Regardless of the alleged huge number of individuals being affected by rare life-threatening conditions, the disease burden estimates of the public health are unreliable. About 50% of the orphan genetic ailments are in kids’ with 30% of them cannot live for more than 5 years.
New-born screening, legislation, and construction of national strategies are the major activities being carried out in the orphan drugs market. Legislation has helped in driving the development of more orphan drugs. For instance, the US congress passed Orphan Drug Act in 1983, and it was mainly intended to encourage development of new drugs for rare diseases. Currently, numerous legislation and strategies are developed to facilitate research of orphan drugs globally in more than 35 countries.
Request For Free Sample @ https://www.kennethresearch.com/sample-request-10078023
Cancer, metabolic diseases, blood diseases, immunologic disease, and neurologic diseases are the types of diseases that are addressed with orphan drugs. Cystic fibrosis, glioma, pancreatic cancer, acute myeloid leukemia, multiple myeloma, renal cell carcinoma, ovarian cancer and Duchenne muscular dystrophy are some of the common indications among the various disease types.
Market Analysis: The “Global Orphan Drugs Market” is estimated to witness a CAGR of 11.9% during the forecast period 2017–2023. The market is analyzed based on three segments, namely types, applications, and regions.
Regional Analysis: The regions covered in the report are North America, Europe, Asia Pacific, and Rest of the World (RoW). North America is the leading region for the orphan drugs market growth followed by Europe. Asia Pacific and RoW are set to be the emerging regions. India, China, and Japan are set to be the most attractive destinations due to the large untapped market.
Types Analysis: The global orphan drugs market by products is segmented into biologics and non-biologics. Biologics orphan drugs is the largest segment in the market with a market share of more than 60% in 2016. It is expected to grow at a double-digit CAGR in the market. However, non-biologic orphan drugs are still expected to hold more than quarter of the market share by 2023. The market is also witnessing various mergers, acquisitions, and collaborations among the top players, which is defining the future of the global orphan drugs market.
Key Players: Novartis AG, F. Hoffmann-La Roche Ltd, Celgene, Bristol-Myers Squibb Company, Shire pharmaceuticals, Pfizer, Sonafi, and Bayer Healthcare are the key players in the market. These top ten players occupy nearly 50% of the orphan drugs market.
Competitive Analysis: Currently, large pharmaceutical companies are extremely active in the orphan drugs market. Merger and acquisition is the major trend in the market. For instance, around 40% of the biotechnology companies are acquired between the year 2008-2012, and they have an orphan drug in development. Novartis, GSK, Roche, and Pfizer are the largest orphan drug companies. Pfizer, Gilead, Roche, Shire, BMY, and Celgene are leading orphan drug acquirers. Around 50% of the top 20 orphan drugs were either acquired or in-licensed by large pharmaceutical companies.
Benefits: The report provides complete details about the usage and adoption rate of orphan drugs during the forecast period and among the regions. With that, key stakeholders can know about the major trends, drivers, investments, vertical player’s initiatives, government initiatives toward the orphan drugs adoption in the upcoming years along with the details of commercial drugs available in the market. Moreover, the report provides details about the major challenges that are going to impact the market growth. Additionally, the report gives complete details about the key business opportunities to key stakeholders to expand their business and capture the revenue in the specific verticals to analyze before investing or expanding the business in this market.
Buy This Premium Report Now @ https://www.kennethresearch.com/report-purchase-id-10078023
Key Stakeholders:
Table of Contents
1 Industry Outlook 10
1.1 Industry Overview 10
1.2 Industry Trends 10
2 Report Outline 12
2.1 Report Scope 12
2.2 Report Summary 12
2.3 Research Methodology 13
2.4 Report Assumptions 13
3 Market Snapshot 15
3.1 Market Definition – Infoholic Research 15
3.2 Segmented Addressable Market (SAM) 15
3.2.1 Advantages of Development of Orphan Drug 16
3.2.2 Trends of orphan drug market 16
3.3 Related Addressable Markets (RAM) 17
3.3.1 Oncology (Cancer) Drugs 17
3.3.2 Active pharmaceutical ingredients (APIs) 18
3.3.3 Over the counter drugs (OTC) 18
4 Market Outlook 20
4.1 Overview 20
4.2 Regulatory Framework for orphan drugs 20
4.3 Funding Scenario for orphan drugs 22
4.4 Market Segmentation 23
4.5 Porter 5(Five) Forces 24
4.6 PEST Analysis 25
5 Market Characteristics 26
5.1 Market Dynamics 26
5.1.1 Drivers 26
Read More @ https://www.kennethresearch.com/report-details/global-orphan-drugs-market/10078023
About Kenneth Research:
Kenneth Research is a reselling agency providing market research solutions in different verticals such as Automotive and Transportation, Chemicals and Materials, Healthcare, Food & Beverage and Consumer Packaged Goods, Semiconductors, Electronics & ICT, Packaging, and Others. Our portfolio includes set of market research insights such as market sizing and market forecasting, market share analysis and key positioning of the players (manufacturers, deals and distributors, etc), understanding the competitive landscape and their business at a ground level and many more. Our research experts deliver the offerings efficiently and effectively within a stipulated time. The market study provided by Kenneth Research helps the Industry veterans/investors to think and to act wisely in their overall strategy formulation
Contact Us
Name: David
Email : Sales@kennethresearch.com
Phone: +1 313 462 0609
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global Orphan Drugs Market will multiply at impressive CAGR of 11.9% by 2024 & Top Key Players are Novartis AG, F. Hoffmann-La Roche Ltd, Celgene, Bristol-Myers Squibb Company, Shire pharmaceuticals and Pfizer here
News-ID: 1680142 • Views: …
More Releases from Kenneth Research
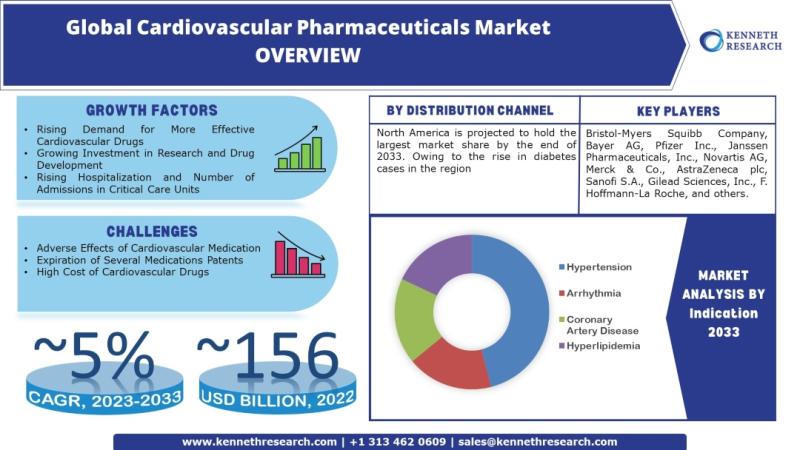
Global Cardiovascular Pharmaceuticals Market to be Propelled by Growing Investme …
Kenneth Research published a report titled "Cardiovascular Pharmaceuticals Market: Global Demand Analysis & Opportunity Outlook 2033" which delivers detailed overview of the global cardiovascular pharmaceuticals market in terms of market segmentation by drug class, distribution channel, indication and by region.
Further, for the in-depth analysis, the report encompasses the industry growth indicators, restraints, supply and demand risk, along with detailed discussion on current and future market trends that are associated with…
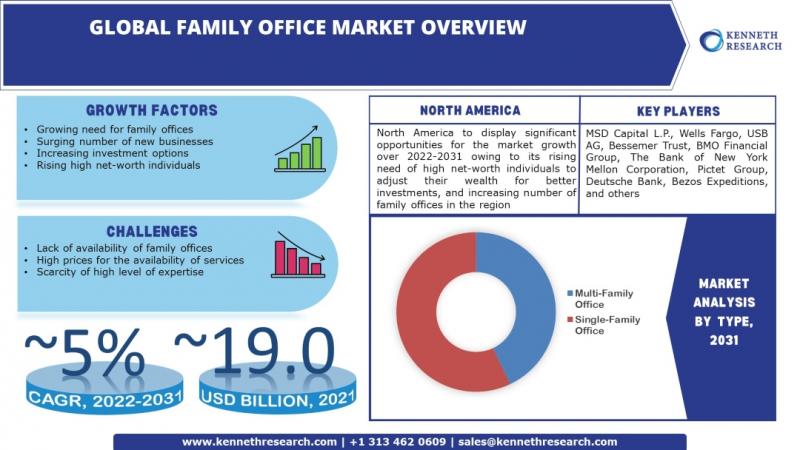
Global Family Office Market to be Propelled by Surging Number of New Businesses …
Kenneth Research published a report titled "Family Office Market: Global Demand Analysis & Opportunity Outlook 2031" which delivers detailed overview of the global family office market in terms of market segmentation by services, type, net worth managed, and by region.
Access Full Description: https://www.kennethresearch.com/report-details/family-office-market/10346745
Further, for the in-depth analysis, the report encompasses the industry growth indicators, restraints, supply and demand risk, along with detailed discussion on current and future market trends that…
Photomask Market Growth Analysis by Revenue, Size, Share, Scenario on Latest Tre …
Kenneth Research provides an extensive study by our analysts which offers forecast assessment by correlating the historical data with key market dynamics. The Photomask Market further includes trends and opportunities that are highlighted, along with the market valuation. The market is segmented by segments and portrays the industry overview along with elaborate description of the market for the forecast period 2020-2025. The report also constitutes future growth statistics which is…
Embedded Systems Market 2020 New Innovations Trends, Research, Global Share and …
Kenneth Research provides an extensive study by our analysts which offers forecast assessment by correlating the historical data with key market dynamics. The Embedded Systems Market further includes trends and opportunities that are highlighted, along with the market valuation. The market is segmented by segments and portrays the industry overview along with elaborate description of the market for the forecast period 2020-2025. The report also constitutes future growth statistics which…
More Releases for Orphan
Acquired Orphan Blood Disease Market
Acquired Orphan Blood Disease Market to reach over USD 18.93 billion by the year 2031 - Exclusive Report by InsightAce Analytic
According to a new report by InsightAce Analytic, the "Acquired Orphan Blood Disease Market" in terms of revenue was estimated to be worth $8.65 billion in 2023 and is poised to reach $18.93 billion by 2031, growing at a CAGR of 10.47% from 2024 to 2031.
Get Free Access to…
Orphan Drugs Market Size to Hit $3199.3 Billion by 2028 | Orphan Drugs Industry …
Market Overview:
According to our experience research team, Orphan Drugs Market was valued at USD 112.36 Billion in 2021, and the global Orphan Drugs industry is projected to reach a value of USD 3199.3 Billion by 2028, at a CAGR of 7.4% during the forecast period 2022-2028
Vantage Market Research is a collection of market research studies on several industries, such as Chemicals, semiconductors & Electronics, Food & Beverages Technology, Energy &…
Orphan Drugs for Cancer Pipeline Analysis
A huge market opportunity is offered by small patient population which suffers from rare or orphan diseases. Among the category of new orphan drugs, Oncology account for the largest disease group in recent years. It has been observed that majority of the orphan drugs in the clinical stages are for rare cancer disease drugs, and are in the late stages of the pipeline. Some of the drugs are being developed…
US Orphan Drug Pipeline Analysis
In recent years, the pharmaceutical industry has been experiencing a paradigm shift. While a large pool of patients was considered as a major source of revenue for pharma companies in the past, the focus is now gradually shifting to small sections of patients suffering from rare disease. In US, this pool of patients is gradually growing and orphan drugs are becoming an extremely attractive business proposition for the pharmaceuticals industry.…
Europe Orphan Drugs Pipeline Analysis
“Europe Orphan Drugs Pipeline Analysis” by PNS Pharma gives comprehensive insight on the various drug profiles under Orphan Drugs status in Europe. Research report covers all the ongoing drug development in various phases. Each drug profiles include detailed information like: Originator, Owner, Collaborator, Technology Provider, Licensee, Development Phase, Development Indications, Mechanism of Action, Chemical Formula, Country of Development and detailed analysis on the development process. The information for particular drug…
Global Orphan Drug Pipeline Analysis
In recent years, the pharmaceutical industry has been experiencing a paradigm shift. While a large pool of patients was considered as a major source of revenue for pharma companies in the past, the focus is now gradually shifting to small sections of patients suffering from rare disease. In US & Europe, this pool of patients is gradually growing and orphan drugs are becoming an extremely attractive business proposition for the…
