Press release
Chronic Kidney Disease Market: Pipeline Review, Developer Landscape and Competitive Insights
Roots Analysis has announced the addition of “Chronic Kidney Disease Market: Pipeline Review, Developer Landscape and Competitive Insights” report to its list of offerings.Ishita Nanda, the principal analyst, stated, “Chronic kidney disease is responsible for approximately 5-10 million deaths per year, worldwide. In fact, over 90% of the patients suffering from reduced kidney function are unaware of their medical condition. The market is therefore, characterized by a pressing need for effective diagnostic and therapeutic solutions.”
The report presents opinions on several key aspects of the market. Among other elements, it includes:
• A detailed assessment of the current market landscape highlighting the drugs available / under development for treatment of chronic kidney disease. Presently, over 160 product candidates are being evaluated across various stages of development for the management / treatment of chronic kidney disease. Of these, 24% are in the preclinical or discovery phase. Amongst the drugs that are under clinical development, 33 are in phase III / pre-registration, 47 in phase II, and 22 in phase I and phase I/II trials. Interestingly, since 2012, 22 product candidates have been commercialized.
• An in-depth analysis of the product pipeline and developer companies, featuring three schematic representations; these include [A] a heptagon representation, highlighting the distribution of the marketed and development stage molecules based on the target clinical condition, [B] a 2X2 grid analysis, representing the distribution of drug candidates across various disease indications, dosing frequencies and stages of development, and [C] a diagrammatic representation of the regional landscape of industry players involved in the development of drugs in this domain, distributed based on the location of their headquarters. Essentially, the market landscape is characterized by the presence of large (31), mid-sized (33) and small-sized companies (47).
• An insightful market assessment summary, highlighting the clinical and commercial attractiveness of pipeline molecules (phase II and phase III), taking into consideration size of enrolled patient population (for the trial in the highest phase of development), route of administration, treatment type (disease modifying agent and drugs offering symptomatic relief) and dosing frequency (for quantifying clinical attractiveness), and target patient population, expected launch date and size of developer company (for quantifying commercial attractiveness).
• An analysis highlighting the key unmet needs across chronic kidney disease, featuring an analysis of content presented on informal (100 patient blogs and over 3,000 social media posts) and formal (180 scientific articles) literary sources, and the opinions expressed by industry experts confirm the existence of several unmet needs within the CKD market.
• An insightful competitive analysis, highlighting the key players in the domain on the basis of the strength of their respective development portfolios, taking into account company size and the stage of development of lead molecules in their respective pipelines.
• A detailed analysis identifying the key opinion leaders (KOLs), featuring a 2X2 analysis to assess the relative experience of certain KOLs who were shortlisted based on their contributions (in terms of involvement in various clinical studies) to this field.
• An analysis of the partnerships that have been established in the recent past. We observed that 28% of the agreements signed between 2015 and 2018 were related to research and development. These were followed by development and commercialization agreements (23%), licensing agreements (22%), commercialization agreements (15%), acquisitions (5%), mergers (5%), and manufacturing agreements (2%).
• An analysis of the investments made at various stages of development in companies that are focused in this area. Across all funding rounds, the most active investors include (in terms of participation in funding rounds) Omega Funds, Flagship Pioneering, Polaris Partners, venBio Select, Oxford Finance, OrbiMed Advisors, Morningside Group, Limulus Venture Partners, Cormorant Asset Management, AJU IB Investment and Sibling Capital.
Nanda further remarked, “The current market landscape is characterized by the presence of established and small-sized companies and prominent academic institutions, which are engaged in the development of various product candidates, either independently or in collaboration with industry players. It is worth mentioning that most of the research activity in this domain is concentrated in the developed regions (North America and Europe).”
The research covers product portfolios of several companies, including:
• Amgen
• Astellas Pharma
• AstraZeneca
• Bayer
• Bristol-Myers Squibb
• Daiichi Sankyo
• Eli Lilly and Company
• GlaxoSmithKline
• Ironwood Pharmaceuticals
• Kyowa Hakko Kirin
• Mitsubishi Tanabe Pharma
• Novartis Pharmaceuticals
• Sanofi
• Shield Therapeutics
• Vifor Pharma
Roots Analysis is a specialist market research company, sharing fresh and independent perspectives in the bio-pharmaceutical industry. The in-depth research, analysis and insights are driven by an experienced leadership team which has gained many years of significant experience in this sector.
For additional details, please visit
https://www.rootsanalysis.com/reports/view_document/chronic-kidney-disease-market-pipeline-review-developer-landscape-and-competitive-insights-/235.html or email sales@rootsanalysis.com
Contact:
Gaurav Chaudhary
+1-604-595-4954
gaurav.chaudhary@rootsanalysis.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Chronic Kidney Disease Market: Pipeline Review, Developer Landscape and Competitive Insights here
News-ID: 1428279 • Views: …
More Releases from Roots Analysis
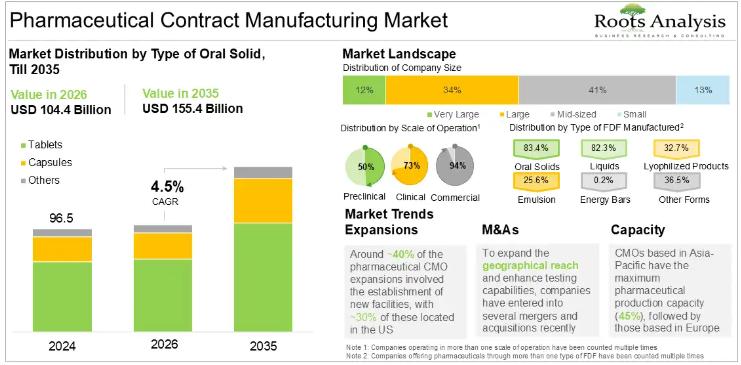
Pharmaceutical Contract Manufacturing Market CAGR To Reach 4.5% between 2025 and …
According to our latest market report "Pharmaceutical Contract Manufacturing Market by Type of Product Manufactured, Type of API, API Potency, Type of FDF, Dosage Form, Type of Oral Solid, Type of Packaging Offered, Scale of Operation, End User, Geographical Regions and Key Players: Industry Trends and Global Forecasts, till 2035", the pharmaceutical contract manufacturing market is estimated to be USD 100.3 billion in 2025. It is expected to reach USD…
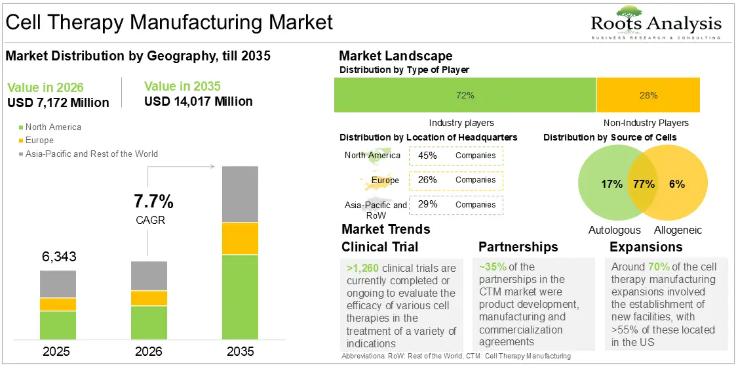
Cell Therapy Manufacturing Market CAGR To Exceed 8.25% by 2035, Due to the Growi …
According to our latest market report "Cell Therapy Manufacturing Market by Type of Cell Therapy, Source of Cells, Scale of Operation, Type of Manufacturer and Key Geographical Regions: Industry Trends and Global Forecasts, 2023-2035", the global cell therapy manufacturing market size is projected to reach USD 14,017 million by 2035 from USD 6,343 million in 2025, growing at a CAGR of 8.25% in the forecast period 2025-2035.
To request quote…
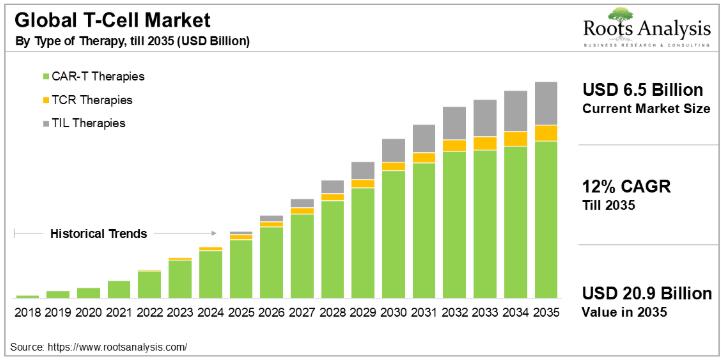
T-Cell Therapy Market Size to Hit USD 20.9 billion by 2035| Exclusive Report by …
Cancer is one of the leading causes of mortality across the world. As per the International Agency for Research on Cancer (IARC), by 2040, there are likely to be 27.5 million new cases and 16.3 million deaths related to cancer, annually. Although cancer therapeutics continue to be one of the most active areas, in terms of drug development, there is still a significant unmet need in this domain. In fact,…
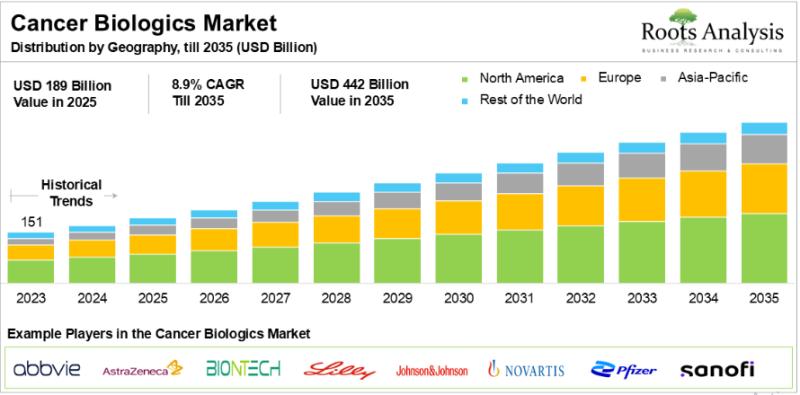
Cancer Biologics Market: Unmet Need and Treatment Guidelines
Owing to the increasing mortality rates and growing need for novel modalities to treat oncological disorders, several researchers and industry stakeholders have shifted their focus on the development of safe and effective biologic therapies. Cancer biologics are the class of therapeutic agents, which primarily modulate immune responses or directly inhibits oncogenic pathways in malignancies. These therapies, such as monoclonal antibodies, specifically target tumor-activating genes, facilitate antibody-dependent cellular cytotoxicity and complement…
More Releases for Pharma
Miglitol Market Size, Share and Forecast By Key Players-Weiao Pharma, Zhejiang M …
𝐔𝐒𝐀, 𝐍𝐞𝐰 𝐉𝐞𝐫𝐬𝐞𝐲- According to the MRI Team's Market Research Intellect, the global Miglitol market is anticipated to grow at a compound annual growth rate (CAGR) of 11.49% between 2024 and 2031. The market is expected to grow to USD 22.32 Billion by 2024. The valuation is expected to reach USD 47.79 Billion by 2031.
The Miglitol market is experiencing significant growth due to the rising prevalence of diabetes and the increasing awareness of the…
Dacarbazine Market Size, Share and Forecast By Key Players-Lingnan Pharma, Ruiyi …
𝐔𝐒𝐀, 𝐍𝐞𝐰 𝐉𝐞𝐫𝐬𝐞𝐲- According to the MRI Team's Market Research Intellect, the global Dacarbazine market is anticipated to grow at a compound annual growth rate (CAGR) of 14.63% between 2024 and 2031. The market is expected to grow to USD 25.32 Billion by 2024. The valuation is expected to reach USD 65.84 Billion by 2031.
The Dacarbazine market is projected to witness significant growth in the coming years, driven by increasing…
Dacarbazine Market Size, Share and Forecast By Key Players-Lingnan Pharma, Ruiyi …
𝐔𝐒𝐀, 𝐍𝐞𝐰 𝐉𝐞𝐫𝐬𝐞𝐲- According to the MRI Team's Market Research Intellect, the global Dacarbazine market is anticipated to grow at a compound annual growth rate (CAGR) of 14.63% between 2024 and 2031. The market is expected to grow to USD 25.32 Billion by 2024. The valuation is expected to reach USD 65.84 Billion by 2031.
The dacarbazine market is experiencing steady growth, driven by the rising incidence of cancer globally. As…
Epilepsy Pipeline Assessment 2024: Therapies, Clinical Trials, and Market Insigh …
(Las Vegas, Nevada, United States) As per DelveInsight's assessment, globally, Epilepsy pipeline constitutes 75+ key companies continuously working towards developing 90+ Epilepsy treatment therapies, analysis of Clinical Trials, Therapies, Mechanism of Action, Route of Administration, and Developments analyzes DelveInsight.
The Epilepsy Pipeline report embraces in-depth commercial and clinical assessment of the pipeline products from the pre-clinical developmental phase to the marketed phase. The report also covers a detailed description of the…
Global Indomethacin Market Seeking Excellent Growth by 2028 | Ratiopharm, Merck, …
"IBI has published a report entitled Global Indomethacin Market Research Report which provides comprehensive data on emerging trends, market drivers, growth opportunities, and barriers that could change the dynamics of the industry market. It provides an in-depth analysis of market segments which includes the product, application, and competitor analysis.
Click Here To Order A Sample Copy Of Indomethacin Global Market Report : https://www.infinitybusinessinsights.com/request_sample.php?id=380382
The Indomethacin Global Market Research Report provides close monitoring…
Roxatidine Market 2020: Top Companies Analysis To Grow Healthcare Business by 20 …
Business Industry Reports Research has recently announced a report on Global Roxatidine Market based on the Category Industry. The Roxatidine Market report emphasizes various key aspects, which include growth drivers, restraints, opportunities and recent market trends for the forecast period 2020-2024.
Global Roxatidine Market overview:
Business Industry Reports Analyst covers the Major Players data, including: shipment, revenue, gross profit, interview record, business distribution etc., these data help the consumer know about the…