Press release
MRI Safe Implantable Defibrillator Devices Market Forecast, Trend Analysis & Competition Tracking - Global Review 2017 to 2027
To study the market for MRI Safe Implantable Defibrillator Devices in a precise manner, Fact.MR has included a new research report. The report is titled “MRI Safe Implantable Defibrillator Devices Market Forecast, Trend Analysis & Competition Tracking - Global Review 2017 to 2027”, which enlightens the readers about the market share as well as growth rate during the period 2018-2027. The research study estimates that the MRI Safe Implantable Defibrillator Devices manufacturing sector would be rising with a positive CAGR during the same stated forecast period.Request TOC of this Report- https://www.factmr.com/connectus/sample?flag=T&rep_id=566
Cardiac defibrillator is a battery powered pulse generator used to correct the abnormal heart rhythms by delivering an electric shock. It is very valuable device in preventing sudden deaths. For several years in the past, MRI was not allowed in patients having implanted devices such as cardiac defibrillator or pacemakers as it may alter pacing properties or even damage myocardium, thus might be raising concerns for clinicians. As, MRI is one of the most useful diagnostic test for patients with various medical problems including cardiology, neurology, orthopaedics etc., therefore the development of MRI safe implantable defibrillator devices helps in solving the problem of risks associated with MRI scans. Several research in the area proves that nearly two-third of patients implanted with a defibrillator needed an MRI diagnosis within four years of implantation, thereby generating the need of MRI safe implantable defibrillator devices.
The MRI safe implantable defibrillator devices are available as single chambered or dual chambered. These devices are either recognized as MR conditional or MR safe devices. They contain specific hardware and software components specifically tested and officially verified for use under magnetic resonance environment.
MRI Safe Implantable Defibrillator Devices Market: Drivers & Restraints
Several product advancements, rising geriatric population, increase in cardiac diseases and number of eligible population are some of the factors supporting the market growth of MRI safe implantable defibrillator devices. Furthermore, rising evidence about safety of these systems and effective marketing campaigns by various manufacturers supports the market growth of MRI safe implantable defibrillator devices. However, high product pricing and stringent regulatory issues for product approval lowers its market growth over the forecast period. Reimbursement issues, lack of skilled professionals in several countries are also limiting the market progress.
Browse Report- https://www.factmr.com/report/566/mri-safe-implantable-defibrillator-devices-market
MRI Safe Implantable Defibrillator Devices Market: Segmentation
Segmentation by Product Type:
Single Chamber Defibrillator
Dual Chamber Defibrillator
Segmentation by Modality:
MR Conditional Defibrillator
MR Safe Defibrillator
Segmentation by End User
Hospitals
Ambulatory Surgical Centers
MRI Safe Implantable Defibrillator Devices Market: Overview
There are very few companies involved in the manufacturing of devices with MRI compatibility. The products are launched in the market only few years back. Medtronic announced the CE Mark and European launch of first MRI safe implantable defibrillator devices to allow for full-body MRI Scans in April 2014. USFDA has also approved the first MRI compatible ICD manufactured by Medtronic recently in September 2015. Despite the launch of MRI safe implantable defibrillator devices, most of the population is still present with the non-MRI compatible devices. Companies in this direction are investing on research and development activities for advancement in these device types to improve therapy outcomes and manufacturing of low cost MRI safe implantable defibrillator devices. Top 3 companies in this market represent most of the market share globally for these MRI safe implantable defibrillator devices due to their strong presence, product approvals, brand strength and lack of competitors.
MRI Safe Implantable Defibrillator Devices Market: Region-wise Outlook
Geographically, North America leads the market for MRI Safe Implantable Defibrillator Devices services owing to the advanced healthcare facilities, high number of implants in the region as well as good reimbursement scenario. This is followed by the Western Europe countries due to easy approval of these devices by CE than by USFDA. Good adoption rate of MRI safe implantable defibrillator devices in the region promotes the growth. APEJ represents a significant growth opportunity for the MRI Safe Implantable Defibrillator devices over the forecast period owing to the rise in cardiac diseases, increase in healthcare spending and rising medical tourism. Japan also represents a substantial market share over the forecast period owing to the high number of geriatric population as well as available government support for medical facilities. Eastern Europe does not represent a significant market share due to less adoption rate of these devices. Latin America followed by Middle East & Africa represents the least market share as well as growth rate in the MRI Safe Implantable Defibrillator devices over the forecast period but represents a significant market opportunity for the market players because of the number of eligible population in these regions.
MRI Safe Implantable Defibrillator Devices Market Treatment Market: Key Players
Some of the players in the MRI Safe Implantable Defibrillator Devices market includes Medtronic, Boston Scientific Corporation, Abbott, Microport, Biotronik etc.
Pre-book Global MRI Safe Implantable Defibrillator Devices Market Forecast Report- https://www.factmr.com/checkout/566/S
About Fact.MR
Fact.MR is a fast-growing market research firm that offers the most comprehensive suite of syndicated and customized market research reports. We believe transformative intelligence can educate and inspire businesses to make smarter decisions. We know the limitations of the one-size-fits-all approach; that’s why we publish multi-industry global, regional, and country-specific research reports.
Contact Us
Fact.MR
Suite 9884
27 Upper Pembroke Street,
Dublin 2, Ireland
Telephone: +353-1-6111-593
Email: sales@factmr.com/
Web: https://www.factmr.com/
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release MRI Safe Implantable Defibrillator Devices Market Forecast, Trend Analysis & Competition Tracking - Global Review 2017 to 2027 here
News-ID: 1009430 • Views: …
More Releases from Fact.MR

Patient Temperature Monitoring Market Overview: Reaching US$ 8.2 Billion by 2026
According to Fact.MR, a market research and competitive intelligence provider, the Global Patient Temperature Monitoring Market is predicted to reach a size of US$ 8.2 billion by 2026, rising at a CAGR of 10% from 2022 to 2026. a patient temperature monitor is a portable gadget that can measure temperature on its own for a brief amount of time. Opportunities for market participants will arise from the large patient pool…
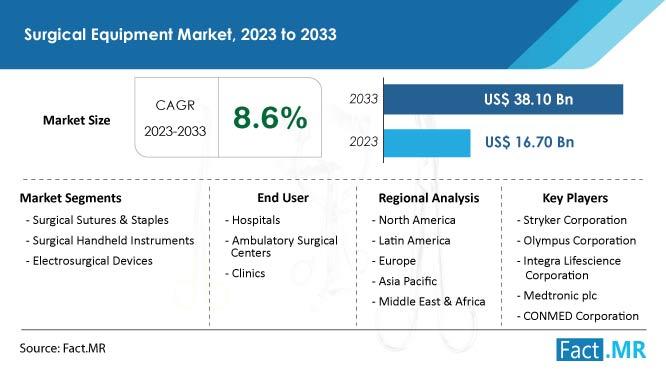
Surgical Equipment Market 8.6% CAGR: The Path to US$ 38.1 Billion By 2033
Fact.MR's global Surgical Equipment Market research study provides a thorough examination of the top market segments according to application, region, business size, solution, and module. It also offers in-depth details on important participants and the primary strategies used to obtain an advantage over rivals. Fact.MR: A Supplier of Competitive Intelligence and Market Research: The surgical equipment market is expected to grow at a remarkable 8.6% CAGR over the following ten…

Tech Support Services Market is Expected to reach US$ 111 Billion by 2033
The global tech support services market is estimated to be valued US$ 66.3 billion in fiscal year 2023, up from US$ 63 billion in FY2022. From 2023 to 2033, the market is predicted to increase at a 5.3% CAGR, reaching $111 billion by the end of 2033.
The market is growing as a result of increased digital technology utilization in a range of end-use industries, as well as rising social media,…

Dairy Stabilizer Market Forecasted to Achieve US$ 134 Million by 2034
The global dairy stabilizer market is expected to generate $81 million in sales in 2024 and grow to $134 million by the end of 2034. Global demand for dairy stabilizers is expected to increase at a 5.1% CAGR over the next ten years.
Dairy stabilizers are special ingredients used in the dairy industry to improve the texture and quality of products like yogurt, ice cream, and cheese. They make sure the…
More Releases for MRI
MRI-guided Neurosurgical Ablation Market Opportunity by 2028 | Insightec Lmt, An …
According to the latest market report published by Future Market Insights titled ‘MRI-Guided Neurosurgical Ablation Market: Global Industry Analysis 2013–2017 and Opportunity Assessment 2018–2028’, the global MRI-guided neurosurgical ablation market is projected to expand at a CAGR of 4.9% during the forecast period 2018–2028 and reach US$ 216.3 Mn by the end of 2028.
Global MRI-guided Neurosurgical Ablation Market: Dynamics
The per capita healthcare expenditure is currently following an uptrend across both…
MAGNETIC RESONANCE IMAGING (MRI) SYSTEM MARKET 2025: BY FIELD STRENGTH (HIGH FIE …
Global Magnetic Resonance Imaging (MRI) System Market Expected to Reach XX Million by 2025
According to a new report published by Reports Monitortitled, “Magnetic Resonance Imaging (MRI) System Market By Architecture & Field Strength: Global Opportunity Analysis and Industry Forecast, 2017–2025,” the Magnetic Resonance Imaging (MRI) system market was valued at XX million in 2016, and is projected to reach at XX million by 2025, growing at a CAGR of XX% from…
Magnetic Resonance Imaging (MRI) Systems Market by Field Strength (High-Field MR …
Global Magnetic Resonance Imaging (MRI) Systems Market was valued at USD 5.63 Billion in the year 2017. Global Magnetic Resonance Imaging (MRI) Systems Market is further estimated to grow at a CAGR of 4.02% from 2018 to reach USD 7.13 Billion by the year 2023. The North America region holds the highest market share in 2018 and the Asia Pacific is considered as the fastest growing market in the forecasted…
Magnetic Resonance Imaging (MRI) Market Global Growth +5.5% CAGR, Architecture ( …
Magnetic Resonance Imaging Devices Market accounted to USD 6.1 billion in 2016 growing at a CAGR of 5.5% during the forecast period of 2017 to 2024. The upcoming market report contains data for historic years 2015, the base year of calculation is 2016 and the forecast period is 2017 to 2024.
Global Magnetic Resonance Imaging (MRI) Market – By Architecture (Closed MRI System, Open MRI System), By Field Strength (Low Field…
Magnetic Resonance Imaging (MRI) Market Growth +5.5% CAGR, Architecture (Closed …
Magnetic Resonance Imaging Devices Market accounted to USD 6.1 billion in 2016 growing at a CAGR of 5.5% during the forecast period of 2017 to 2024. The upcoming market report contains data for historic years 2015, the base year of calculation is 2016 and the forecast period is 2017 to 2024.
Free Sample Report of Global Magnetic Resonance Imaging (MRI) Market @ http://databridgemarketresearch.com/request-a-sample/?dbmr=global-magnetic-resonance-imaging-mri-market
Global Magnetic Resonance Imaging (MRI) Market – By Architecture…
Market Application Of Global MRI Guided Neurosurgical Ablations, Growth Elements …
Qyresearchreports include new market research report "Global MRI Guided Neurosurgical Ablations Market Size, Status and Forecast 2022" to its huge collection of research reports.
Every key element that has happened so far in the MRI Guided Neurosurgical Ablations has been represented in this widely inclusive report. A key portion of these market elements incorporate the key player profiles and the market's general competitive scene, as players are known to have what's…
